2-丙基四氢吡喃 | 3857-17-8
中文名称
2-丙基四氢吡喃
中文别名
2-正丙基四氢吡喃
英文名称
2-propyltetrahydropyran
英文别名
2-n-Propyl-tetrahydropyran;2-propyloxane
CAS
3857-17-8
化学式
C8H16O
mdl
MFCD00049218
分子量
128.214
InChiKey
YHQBBDKFBJYMPJ-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:152-153 °C
-
密度:0.860 g/cm3(Temp: 15 °C)
-
保留指数:958
-
稳定性/保质期:
在常温常压下保持稳定
计算性质
-
辛醇/水分配系数(LogP):2.2
-
重原子数:9
-
可旋转键数:2
-
环数:1.0
-
sp3杂化的碳原子比例:1.0
-
拓扑面积:9.2
-
氢给体数:0
-
氢受体数:1
安全信息
-
储存条件:常温、避光、通风干燥处密封保存。
SDS
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2-丙基-四氢-吡喃-4-醇 2-propyloxan-4-ol 23077-46-5 C8H16O2 144.214 4-(四氢-呋喃-2-基)-2-丁醇 4-tetrahydrofuran-2-yl-butan-2-ol 4527-76-8 C8H16O2 144.214 4-氯-2-丙基-四氢-吡喃 4-chloro-2-propyl-tetrahydro-pyran 53045-60-6 C8H15ClO 162.66 1-甲氧基辛烷-5-醇 1-methoxyoctan-5-ol 53118-76-6 C9H20O2 160.257
反应信息
-
作为反应物:参考文献:名称:Bel'skii; Schuikin, Doklady Akademii Nauk SSSR, 1959, vol. 127, p. 91摘要:DOI:
-
作为产物:描述:5-辛烯-1-醇 在 3-butyl-1-(butyl-4-sulfobutyl)imidazolium trifluoromethanesulfonate 作用下, 以 苯 为溶剂, 反应 93.0h, 以39%的产率得到2-丙基四氢吡喃参考文献:名称:布朗斯台德酸性离子液体中的分子内加氢烷氧基化及其在合成(±)-中心洛宾中的应用†摘要:SO 3 H系咪唑鎓盐和三唑鎓盐,不挥发且可回收的布朗斯台德酸性离子液体,可有效介导链烯醇的分子内加氢烷氧基化反应。它们已成功地用于合成(±)-中心洛宾。DOI:10.1039/c0ob00701c
文献信息
-
SUBSTITUTED 4-PYRIDONES AND THEIR USE AS INHIBITORS OF NEUTROPHIL ELASTASE ACTIVITY申请人:OOST Thorsten公开号:US20140057916A1公开(公告)日:2014-02-27This invention relates to substituted 4-pyridones of formula 1 and their use as inhibitors of neutrophil elastase activity, pharmaceutical compositions containing the same, and methods of using the same as agents for treatment and/or prevention of pulmonary, gastrointestinal and genitourinary diseases, inflammatory diseases of the skin and the eye and other auto-immune and allergic disorders, allograft rejection, and oncological diseases.
-
8-AMINOISOQUINOLINE COMPOUNDS AND USES THEREOF申请人:Genentech, Inc.公开号:US20200108075A1公开(公告)日:2020-04-093-Carbonylamino-8-aminoisoquinoline compounds of formula (I): variations thereof, and their use as inhibitors of HPK1 (hematopoietic kinase 1) are described. The compounds are useful in treating HPK1-dependent disorders and enhancing an immune response. Also described are methods of inhibiting HPK1, methods of treating HPK1-dependent disorders, methods for enhancing an immune response, and methods for preparing the 3-carbonylamino-8-aminoisoquinoline compounds.
-
[EN] N-PYRIDINYL ACETAMIDE DERIVATIVES AS INHIBITORS OF THE WNT SIGNALLING PATHWAY<br/>[FR] DÉRIVÉS DE N-PYRIDYL ACÉTAMIDE UTILISÉS COMME INHIBITEURS DE LA VOIE DE SIGNALISATION WNT申请人:REDX PHARMA PLC公开号:WO2016055786A1公开(公告)日:2016-04-14This invention relates to compounds. More specifically, the invention relates to compounds useful as inhibitors of the Wnt signalling pathway. Specifically, inhibitors of Porcupine (Porcn) are contemplated by the invention. In addition the invention contemplates processes to prepare the compounds and uses of the compounds. The compounds of the invention may therefore be used in treating conditions mediated by the Wnt signalling pathway, for example treating cancer, sarcoma, melanoma, skin cancer, haematological tumors, lymphoma, carcinoma, and leukemia; or enhancing the effectiveness of an anti-cancer treatment.
-
Compounds, pharmaceutical compositions and uses thereof申请人:HIMMELSBACH Frank公开号:US20120322784A1公开(公告)日:2012-12-20The present invention relates to compounds of formula I, wherein the groups R 1 , L P , L Q , X 1 , X 2 , X 3 , Ar and n are as defined in the application, which have valuable pharmacological properties, and in particular bind to the GPR119 receptor and modulate its activity.本发明涉及公式I的化合物, 其中R 1 ,LP,LQ,X 1 ,X 2 ,X 3 ,Ar和n的定义如申请中所述,具有有价值的药理特性,特别是结合GPR119受体并调节其活性。
-
Reaction of Acetals with Grignard Reagents作者:Hiroshi Ishikawa、Teruaki Mukaiyama、Shigeru IkedaDOI:10.1246/bcsj.54.776日期:1981.3The reaction of dialkyl acetals derived from α,β-unsaturated aldehydes with Grignard reagents using TiCl4in THF afforded the cross coupling products, allyl ethers, in high yields. The TiCl4-promoted reaction of alkyl 2,4-dichlorophenyl acetals, synthesized from 3,4-dihydro-2H-pyran or ethyl vinyl ether and 2,4-dichlorophenol, with Grignard reagents in THF at low temperature afforded the corresponding
表征谱图
-
氢谱1HNMR
-
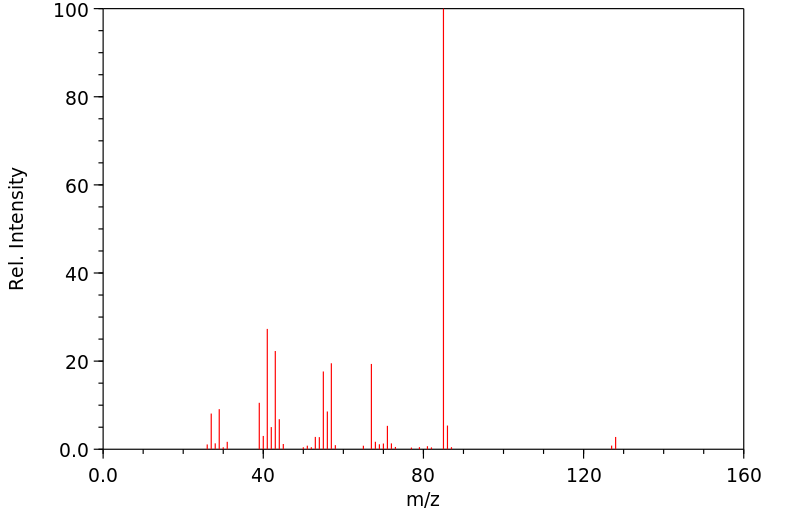
质谱MS
-
碳谱13CNMR
-
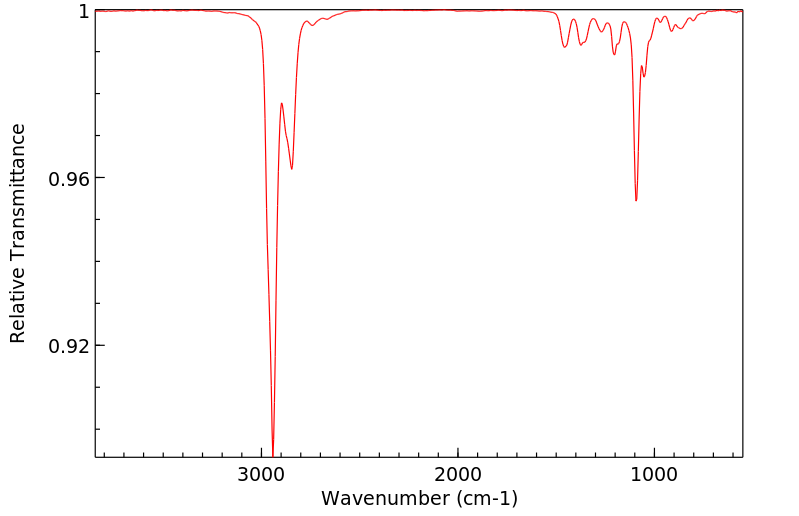
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(3S,4R)-3-氟四氢-2H-吡喃-4-胺
鲁比前列素中间体
顺式-3-溴<2-(2)H>四氢吡喃
顺-4-氨基四氢吡喃-3-醇
顺-4-(四氢吡喃-2-氧)-2-丁烯-1-醇
顺-3-Boc-氨基-四氢吡喃-4-羧酸
锡烷,三丁基[3-[(四氢-2H-吡喃-2-基)氧代]-1-炔丙基]-
螺[金刚烷-2,2'-四氢吡喃]-4'-醇
蒿甲醚四氢呋喃乙酸酯
蒜味伞醇B
蒜味伞醇A
茉莉吡喃
苯基2,4-二氯-5-氨磺酰苯磺酸酯
苄基2,3-二-O-乙酰基-4-脱氧-4-C-硝基亚甲基-β-D-阿拉伯吡喃果糖苷
膜质菊内酯
红没药醇氧化物A
红没药醇氧化物
科立内酯
硅烷,(1,1-二甲基乙基)二甲基[[4-[(四氢-2H-吡喃-2-基)氧代]-5-壬炔基]氧代]-
甲磺酸酯-四聚乙二醇-四氢吡喃醚
甲基[(噁烷-3-基)甲基]胺
甲基6-氧杂双环[3.1.0]己烷-2-羧酸酯
甲基4-脱氧吡喃己糖苷
甲基3-脱氧-3-硝基-beta-L-核吡喃糖苷
甲基2,4,6-三脱氧-2,4-二-C-甲基吡喃葡己糖苷
甲基1,2-环戊烯环氧物
甲基-[2-吡咯烷-1-基-1-(四氢-吡喃-4-基)-乙基]-胺
甲基-(四氢吡喃-4-甲基)胺
甲基-(四氢吡喃-2-甲基)胺盐酸盐
甲基-(四氢吡喃-2-甲基)胺
甲基-(四氢-吡喃-3-基-胺
甲基-(四氢-吡喃-3-基)-胺盐酸盐
甲基-(4-吡咯烷-1-甲基四氢吡喃-4-基)-胺
甲基(5R)-3,4-二脱氧-4-氟-5-甲基-alpha-D-赤式-吡喃戊糖苷
环氧乙烷-2-醇乙酸酯
环己酮,6-[(丁基硫代)亚甲基]-2,2-二甲基-3-[(四氢-2H-吡喃-2-基)氧代]-,(3S)-
环丙基-(四氢-吡喃-4-基)-胺
玫瑰醚
独一味素B
溴-六聚乙二醇-四氢吡喃醚
氯菊素
氯丹环氧化物
氨甲酸,[[(四氢-2H-吡喃-2-基)氧代]甲基]-,乙基酯
氨甲酸,[(4-氨基四氢-2H-吡喃-4-基)甲基]-,1,1-二甲基乙基酯(9CI)
氧杂-3-碳酰肼
氧化氯丹
正-(四氢-4-苯基-2h-吡喃-4-基)乙酰胺
次甲霉素 A
桉叶油醇
无