3-甲基喹唑啉-2,4(1h,3h)-二酮 | 607-19-2
中文名称
3-甲基喹唑啉-2,4(1h,3h)-二酮
中文别名
3-甲基喹唑啉-2,4(1H,3H)-二酮
英文名称
3-methyl-2,4(1H,3H)-quinazolinedione
英文别名
3-methylquinazoline-2,4(1H,3H)-dione;3-methyl-1H-quinazoline-2,4-dione;1,2,3,4-tetrahydro-3-methyl-2,4-dioxo-quinazoline
CAS
607-19-2
化学式
C9H8N2O2
mdl
MFCD01534101
分子量
176.175
InChiKey
LFGZKCYBINPOTA-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:236-238 °C
-
密度:1.292±0.06 g/cm3(Predicted)
计算性质
-
辛醇/水分配系数(LogP):0.6
-
重原子数:13
-
可旋转键数:0
-
环数:2.0
-
sp3杂化的碳原子比例:0.111
-
拓扑面积:49.4
-
氢给体数:1
-
氢受体数:2
安全信息
-
海关编码:2933990090
SDS
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2,4-喹唑啉二酮 1,2,3,4-tetrahydro-quinazoline-2,4-dione 86-96-4 C8H6N2O2 162.148 2-疏基-3-甲基-3H-喹唑啉-4-酮 3-methyl-4-oxoquinazoline-2-thione 1705-09-5 C9H8N2OS 192.241 —— 3-Methyl-4-oxo-3,4-dihydro-quinazoline-2-carbonitrile 213211-95-1 C10H7N3O 185.185 —— (3-methyl-4-oxo-3,4-dihydroquinazolin-2-ylsulfanyl)acetic acid ethyl ester —— C13H14N2O3S 278.332 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 5-chloro-3-methyl-2,4(1H,3H)-quinazolinedione 118470-98-7 C9H7ClN2O2 210.62 —— 1-(3-chloropropyl)-2,4-dioxo-3-methyl-1,2,3,4-tetrahydroquinazoline 221540-05-2 C12H13ClN2O2 252.7 —— 1-(4-chlorobutyl)-2,4-dioxo-3-methyl-1,2,3,4-tetrahydroquinazoline 221540-06-3 C13H15ClN2O2 266.727 —— 3-methyl-1-phenethyl-3,4-dihydroquinazolin-2(1H,3H)-dione —— C17H16N2O2 280.326 —— 1-trichloromethylthio-3-methyl-2,4-dioxo-1,3-dihydroquinazoline 88634-92-8 C10H7Cl3N2O2S 325.603 —— 3-Methyl-1-(3-methylsulfanylphenyl)quinazoline-2,4-dione 1196453-25-4 C16H14N2O2S 298.365
反应信息
-
作为反应物:描述:参考文献:名称:Abt, Journal fur praktische Chemie (Leipzig 1954), 1889, vol. <2> 39, p. 142摘要:DOI:
-
作为产物:描述:参考文献:名称:Synthesis and evaluation of highly selective quinazoline-2,4-dione ligands for sphingosine-1-phosphate receptor 2摘要:新的喹唑啉-2,4-二酮类似物被开发为鞘氨醇1磷酸受体2配体。[11C]2i 在外周组织疾病中作为正电子发射断层扫描探针具有巨大潜力。DOI:10.1039/d1md00357g
文献信息
-
Synthesis and characterization of the first inhibitor of<i>N</i>-acylphosphatidylethanolamine phospholipase D (NAPE-PLD)作者:Beatrice Castellani、Eleonora Diamanti、Daniela Pizzirani、Piero Tardia、Martina Maccesi、Natalia Realini、Paola Magotti、Gianpiero Garau、Thomas Bakkum、Silvia Rivara、Marco Mor、Daniele PiomelliDOI:10.1039/c7cc07582k日期:——(NAPE-PLD) is a membrane-associated zinc enzyme that catalyzes the hydrolysis of N-acylphosphatidylethanolamines (NAPEs) into fatty acid ethanolamides (FAEs). Here, we describe the identification of the first small-molecule NAPE-PLD inhibitor, the quinazoline sulfonamide derivative 2,4-dioxo-N-[4-(4-pyridyl)phenyl]-1H-quinazoline-6-sulfonamide, ARN19874.
-
QUINAZOLINE-2,4-DIONE DERIVATIVES申请人:Hubschwerlen Christian公开号:US20140171425A1公开(公告)日:2014-06-19The invention relates to antibacterial compounds of formula (I), wherein R 1 is H, halogen, (C 1 -C 3 )alkyl or (C 1 -C 3 )alkoxy; R 2 is H, halogen, (C 1 -C 3 )alkyl, (C 1 -C 3 )alkoxy or pyrrolidin-1-yl; R 3 is H, halogen, (C 1 -C 3 )alkyl, (C 1 -C 3 )alkoxy, vinyl or 2-methoxycarbonyvinyl or R 2 and R 3 together with the two carbon atoms which bear them form a phenyl ring; R 4 is H, halogen, (C 1 -C 3 )alkyl or (C 1 -C 3 )alkoxy; and R 5 is H, (C 1 -C 3 )alkyl or cyclopropyl, or R 4 and R 5 form together a —CH 2 CH 2 CH 2 — group; A is the divalent group —CH 2 —, —CH 2 CH 2 —, #—CH(OH)CH 2 —*, #—CH 2 N(R 6 )—* and —CH 2 NHCH 2 —, wherein # indicates the point of attachment to the optionally substituted (quinazoline-2,4-dione-3-yl)methyl residue and * represents the point of attachment to the substituted (oxazolidinon-4-yl)methyl residue; R 6 is H or acetyl; Y is CH or N; and Q is O or S; and salts of such compounds.该发明涉及式(I)的抗菌化合物,其中R1为H、卤素、(C1-C3)烷基或(C1-C3)氧烷;R2为H、卤素、(C1-C3)烷基、(C1-C3)氧烷或吡咯烷-1-基;R3为H、卤素、(C1-C3)烷基、(C1-C3)氧烷、乙烯基或2-甲氧羰基乙烯基,或R2和R3与携带它们的两个碳原子一起形成苯环;R4为H、卤素、(C1-C3)烷基或(C1-C3)氧烷;R5为H、(C1-C3)烷基或环丙基,或R4和R5一起形成一个— —基团;A为二价基团—CH2—、— —、#—CH(OH) —*、#— N(R6)—*和— NH —,其中#表示可选择取代的(喹唑啉-2,4-二酮-3-基)甲基残基的连接点,*表示取代的(噁唑烷酮-4-基)甲基残基的连接点;R6为H或乙酰基;Y为CH或N;Q为O或S;以及这类化合物的盐。
-
[EN] QUINAZOLINE-2,4-DIONE DERIVATIVES<br/>[FR] DÉRIVÉS DE QUINAZOLINE-2,4-DIONE申请人:ACTELION PHARMACEUTICALS LTD公开号:WO2013021363A1公开(公告)日:2013-02-14The invention relates to antibacterial compounds of formula (I), wherein R1 is H, halogen, (C1-C3)alkyl or (C1-C3)alkoxy; R2 is H, halogen, (C1-C3)alkyl, (C1-C3)alkoxy or pyrrolidin-l-yl; R3 is H, halogen, (C1-C3)alkyl, (C1-C3)alkoxy, vinyl or 2-methoxycarbonylvinyl or R2 and R3 together with the two carbon atoms which bear them form a phenyl ring; R4 is H, halogen, (C1-C3)alkyl or (C1-C3)alkoxy and R5 is H, (C1-C3)alkyl or cyclopropyl, or R4 and R5 form together a -CH2CH2CH2- group; A is the divalent group -CH2-, -CH2CH2-, #-CH(OH)CH2-*, #-CH2N(R6)-* or -CH2NHCH2-, wherein # indicates the point of attachment to the optionally substituted (quinazoline-2,4-dione-3-yl)methyl residue and * represents the point of attachment to the substituted (oxazolidinon-4-yl)methyl residue; R6 is H or acetyl; Y is CH or N; and Q is O or S; and salts of such compounds.该发明涉及式(I)的抗菌化合物,其中R1为H、卤素、(C1-C3)烷基或(C1-C3)氧烷基;R2为H、卤素、(C1-C3)烷基、(C1-C3)氧烷基或吡咯烷-1-基;R3为H、卤素、(C1-C3)烷基、(C1-C3)氧烷基、乙烯基或2-甲氧羰基乙烯基,或者R2和R3与它们所连接的两个碳原子一起形成苯环;R4为H、卤素、(C1-C3)烷基或(C1-C3)氧烷基,R5为H、(C1-C3)烷基或环丙基,或者R4和R5一起形成一个- -基团;A为二价基团-CH2-、- -、#-CH(OH) -*、#- N(R6)-*或- NH -,其中#表示可选择取代的(喹唑啉-2,4-二酮-3-基)甲基残基的连接点,*表示取代的(噁唑烷酮-4-基)甲基残基的连接点;R6为H或乙酰基;Y为CH或N;Q为O或S;以及这类化合物的盐。
-
[EN] 2,4-DIOXO-QUINAZOLINE-6-SULFONAMIDE DERIVATIVES AS INHIBITORS OF PARG<br/>[FR] DÉRIVÉS DE 2,4-DIOXO-QUINAZOLINE-6-SULFONAMIDE EN TANT QU'INHIBITEURS DE LA PARG申请人:CANCER REC TECH LTD公开号:WO2016092326A1公开(公告)日:2016-06-16The present invention relates to compounds of formula I that function as inhibitors of PARG (Poly ADP-ribose glycohydrolase) enzyme activity wherein R1a, R1b, R1c, R1d, R1e, W, X1, X2, X3, X4, X5, X6, X7, c are each as defined herein. The present invention also relates to processes for the preparation of these compounds, to pharmaceutical compositions comprising them, and to their use in the treatment of proliferative disorders, such as cancer, as well as other diseases or conditions in which PARG activity is implicated.
-
Cell-Active Small Molecule Inhibitors of the DNA-Damage Repair Enzyme Poly(ADP-ribose) Glycohydrolase (PARG): Discovery and Optimization of Orally Bioavailable Quinazolinedione Sulfonamides作者:Bohdan Waszkowycz、Kate M. Smith、Alison E. McGonagle、Allan M. Jordan、Ben Acton、Emma E. Fairweather、Louise A. Griffiths、Niall M. Hamilton、Nicola S. Hamilton、James R. Hitchin、Colin P. Hutton、Dominic I. James、Clifford D. Jones、Stuart Jones、Daniel P. Mould、Helen F. Small、Alexandra I. J. Stowell、Julie A. Tucker、Ian D. Waddell、Donald J. OgilvieDOI:10.1021/acs.jmedchem.8b01407日期:2018.12.13DNA damage repair enzymes are promising targets in the development of new therapeutic agents for a wide range of cancers and potentially other diseases. The enzyme poly(ADP-ribose) glycohydrolase (PARG) plays a pivotal role in the regulation of DNA repair mechanisms; however, the lack of potent drug-like inhibitors for use in cellular and in vivo models has limited the investigation of its potential
表征谱图
-
氢谱1HNMR
-
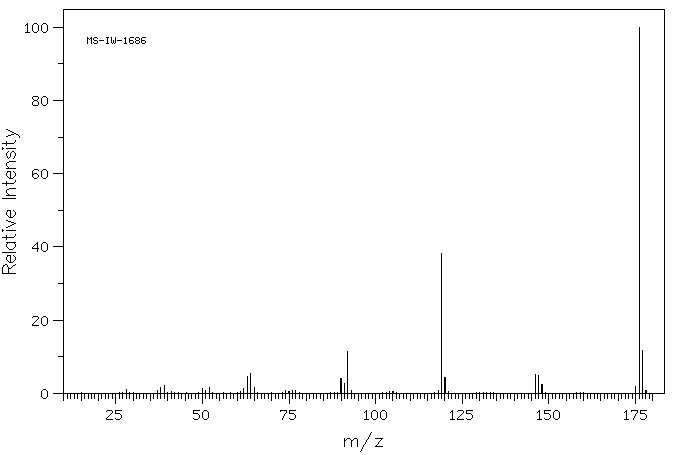
质谱MS
-
碳谱13CNMR
-
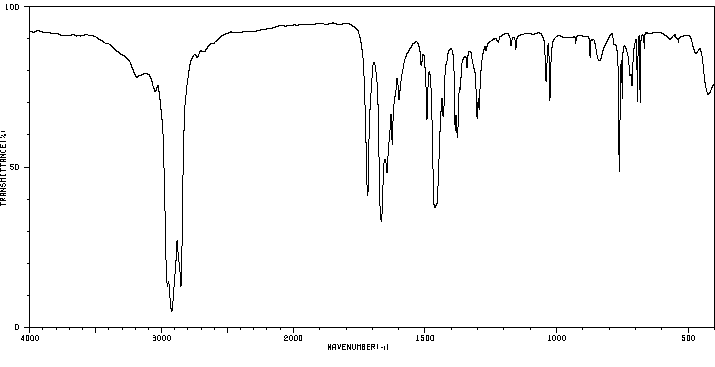
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(12羟基吲[2,1-b〕喹唑啉-6(12H)-酮)
黑暗猝灭剂BHQ-3,BHQ-3NHS
鸭嘴花酚碱
鸭嘴花碱酮;(S)-2,3-二氢-3,7-二羟基吡咯并[2,1-b]喹唑啉-9(1H)-酮
鸭嘴花碱酮
鸭嘴花碱盐酸盐
鲁米诺单钠盐
鲁米诺
骆驼蓬碱
颜料蓝64
颜料蓝60
顺式-卤夫酮
顺式-(喹喔啉-2-基)丙烯腈1,4-二氧化物
非奈利酮
青黛酮
雷替曲塞杂质1
阿法替尼杂质J
阿法替尼杂质I
阿法替尼杂质28
阿法替尼杂质18
阿法替尼杂质13
阿法替尼杂质
阿法替尼中间体
阿法替尼
阿法替尼
阿朴藏红
阿巴康唑
阿夫唑嗪杂质A
阿夫唑嗪杂质
阿夫唑嗪EP杂质C
阿夫唑嗪
阿喹司特
阿呋唑嗪杂质
阿呋唑嗪杂质
铜迈星
铁诱导细胞死亡激活剂
钠四丙基硼酸酯
酸性蓝98
酸性红101
酮色林醇
酞联氮基[2,3-b]酞嗪-5,14-二酮,7,12-二氢-
酞嗪-5-羧酸
酞嗪-2-氧化物
酚藏花红
酚嗪
酒石酸溴莫尼定
邻苯二甲酰肼
还原黄6GD
还原蓝6
达尼喹酮