5-(二乙氨基)-2-(亚硝基苯酚) | 6358-20-9
中文名称
5-(二乙氨基)-2-(亚硝基苯酚)
中文别名
——
英文名称
5-diethylamino-2-nitrosophenol
英文别名
3-hydroxy-4-nitroso-N,N-diethylaniline;5-(Diethylamino)-2-nitrosophenol
CAS
6358-20-9
化学式
C10H14N2O2
mdl
MFCD00009980
分子量
194.233
InChiKey
NXYYRSJPSPRDEO-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:358.1±32.0 °C(Predicted)
-
密度:1.12±0.1 g/cm3(Predicted)
计算性质
-
辛醇/水分配系数(LogP):2.3
-
重原子数:14
-
可旋转键数:3
-
环数:1.0
-
sp3杂化的碳原子比例:0.4
-
拓扑面积:52.9
-
氢给体数:1
-
氢受体数:4
SDS
制备方法与用途
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 3-羟基-N,N-二乙基苯胺 3-diethylaminophenol 91-68-9 C10H15NO 165.235 N,N-二乙基-4-亚硝基苯胺 N,N-diethyl-4-nitrosoaniline 120-22-9 C10H14N2O 178.234 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 2-amino-5-(diethylamino)phenol 17626-05-0 C10H16N2O 180.25
反应信息
-
作为反应物:描述:5-(二乙氨基)-2-(亚硝基苯酚) 在 hydrazine hydrate 、 palladium on activated charcoal 作用下, 以 乙醇 为溶剂, 生成 2-amino-5-(diethylamino)phenol参考文献:名称:一种用于检测二氧化硫衍生物的比率型近红外荧光探针及其制备方法摘要:本发明属于荧光检测技术领域,具体涉及一种用于检测二氧化硫(SO2)衍生物的比率型近红外荧光探针及其制备方法,以及在检测农作物叶片组织中SO2衍生物、大气中的SO2衍生物或土壤中SO2衍生物方面的应用。本发明以间二乙基氨基苯酚、亚硝酸钠、水合肼、钯碳催化剂、丙酮酸乙酯、二氧化硒和1‑乙基‑2‑甲基喹啉等为原料,制备用于检测二氧化硫衍生物的比率型近红外荧光探针,并将其用于检测大气中的SO2衍生物、土壤中SO2衍生物或农作物叶片组织中SO2衍生物。该荧光探针合成方法简单,对SO2衍生物有很好的选择性,灵敏度高,检测环境适用于大范围的pH。尝试的探针试纸是一种简单、便于携带的工具,可以不需要依赖大型仪器设备,而实现对大气中二氧化硫衍生物的快速现场检测,因此具有革新性意义。公开号:CN111825665A
-
作为产物:描述:参考文献:名称:取代的9-二乙基氨基苯并[ a ]苯恶嗪-5-酮(尼罗红类似物):合成和光物理性质摘要:尼罗红是在9位含有二乙氨基取代基的苯并[ ]苯恶a酮染料。近年来,由于其在亲脂性环境中无与伦比的荧光特性,它已成为细胞膜和脂滴的流行组织学染色剂。这使其成为化学装饰的诱人线索,可以调整其属性并针对更专门的显微镜技术对其进行优化,例如荧光寿命成像或双光子激发荧光显微镜,而尼罗红从未对其进行优化。在这里,我们提出了一系列单取代的尼罗红衍生物(9-二乙基苯并[ a][phenoxazin-5-ones)起始于1-萘酚或1,3-萘二醇。报道了这些荧光团的溶剂变色响应性,重点是取代基如何影响吸收和发射光谱,发光度,荧光寿命和双光子吸收率。一些类似物成为报告其当地环境极性的强有力候选者。具体来说,尼罗红的单光子和双光子激发荧光证明对取代反应非常敏感,可以通过在特定位置明智地引入具有不同电子特性的取代基来精细调整光谱特征。这个新的9-二乙基苯并[ a]工具包] phenoxazine-5-ones迈向了DOI:10.1021/acs.joc.0c02346
文献信息
-
Superoxide Radical Photogenerator with Amplification Effect: Surmounting the Achilles’ Heels of Photodynamic Oncotherapy作者:Mingle Li、Tao Xiong、Jianjun Du、Ruisong Tian、Ming Xiao、Lianying Guo、Saran Long、Jiangli Fan、Wen Sun、Kun Shao、Xiangzhi Song、James W. Foley、Xiaojun PengDOI:10.1021/jacs.8b13141日期:2019.2.13transfer theory, the energy donor successfully endows ENBOS with significantly enhanced NIR absorbance and photon utility, which in turn lead to ENBOS more easily activated and generating more O2-• in deep tissues, that thus dramatically intensifies the type I PDT against hypoxic deep tumors. Moreover, benefiting from the dyad cationic feature, ENBOS achieves superior "structure-inherent targeting"氧依赖强、肿瘤靶向性差、治疗深度有限被认为是光动力疗法(PDT)临床应用面临的“致命弱点”。与常见方法不同,在这里,我们提出了一种创新策略,通过使用具有“0 + 1 > 1”放大效应的光子引发的二元阳离子超氧化物自由基 (O2-•) 发生器 (ENBOS) 同时克服这些缺点。特别是,通过利用 Förster 共振能量转移理论,能量供体成功地赋予 ENBOS 显着增强的 NIR 吸收和光子效用,进而导致 ENBOS 更容易激活并在深层组织中产生更多的 O2-•,从而显着增强了针对缺氧深部肿瘤的 I 型 PDT。而且,受益于二元阳离子特征,ENBOS 实现了卓越的“结构固有靶向”能力,静脉注射后 48 小时的信噪比高达 25.2,为精确成像引导的肿瘤治疗提供了机会。同时,瘤内积累和保留性能也显着改善(> 120 h)。基于这些独特的优点,ENBOS 在低光剂量照射下选择性地抑制深部缺氧肿瘤增殖
-
BODIPY/Nile-Red-Based Efficient FRET Pair: Selective Assay of Endoplasmic Reticulum Membrane Fluidity作者:Zhigang Yang、Youngjin Wi、Young-Min Yoon、Peter Verwilst、Joo Hee Jang、Tae Woo Kim、Chulhun Kang、Jong Seung KimDOI:10.1002/asia.201501060日期:2016.2We synthesized a boron‐dipyrromethene (BODIPY)/Nile Red hybrid probe capable of selectively recognizing fluidity changes in the endoplasmic reticulum (ER) membrane due to its preferential localization to the ER and strong energy transfer from BODIPY to the Nile Red moiety, emitting only in nonaqueous environments. ER membrane fluidity in HepG2 cells was markedly reduced by a cell model of metabolic
-
A General Method To Increase Stokes Shift by Introducing Alternating Vibronic Structures作者:Tian-Bing Ren、Wang Xu、Wei Zhang、Xing-Xing Zhang、Zhi-Yao Wang、Zhen Xiang、Lin Yuan、Xiao-Bing ZhangDOI:10.1021/jacs.8b04404日期:2018.6.20common fluorophores. By simply appending a 1,4-diethyl-decahydro-quinoxaline (DQ) moiety onto the conjugated structure, we introduced a vibronic backbone that could facilely expand the Stokes shifts, emission wavelength, and photostability of 11 different fluorophores by more than 3-fold. This generalizable method could significantly improve the imaging efficiency of commercial fluorophores. As a demonstration荧光染料在广泛的生物医学领域取得了很大进展。然而,许多市售染料都存在小的斯托克斯位移,导致当前显微镜配置的信噪比和自淬火较差。在这项工作中,我们开发了一种通用方法来显着增加常见荧光团的斯托克斯位移。通过简单地将 1,4-二乙基-十氢-喹喔啉 (DQ) 部分附加到共轭结构上,我们引入了一个振动骨架,可以轻松地将 11 种不同荧光团的斯托克斯位移、发射波长和光稳定性扩大 3 倍以上. 这种可推广的方法可以显着提高商业荧光团的成像效率。作为示范,我们表明半花青的 DQ 衍生物在小鼠模型中产生了超过吲哚菁绿的 5 倍信号。此外,DQ 修饰的荧光团可以与其母分子配对进行单激发、多发射成像,使我们能够更稳健地研究细胞行为。这种方法有望产生适用于超分辨率显微镜和第二窗口近红外成像的染料。
-
Highly Functionalized β-Cyclodextrins by Solid-Supported Synthesis作者:Nesrin Vurgun、Mark NitzDOI:10.1002/chem.201800028日期:2018.3.20immobilization of the mono‐thiol functionalized β‐CD on PEGA resin via a disulfide bond, enabling solid‐phase elaboration of the remaining six primary amines. To showcase the potential of this method, the amines were elaborated to tripeptides through standard Fmoc‐peptide chemistry. A small library of CD‐tripeptide conjugates was generated which, when reduced from the solid support, could be tagged at the
-
Amorphous Amides申请人:Xerox Corporation公开号:US20150105587A1公开(公告)日:2015-04-16An amorphous amide compound of the formula wherein R is selected from the group consisting of an alkyl group, an aryl group, an alkylaryl group, an arylalkyl group, and combinations thereof. An amorphous diamide compound of the formula wherein R 1 is selected from the group consisting of an alkylene group, an arylene group, an alkylarylene group, an arylalkylene group, and combinations thereof.
表征谱图
-
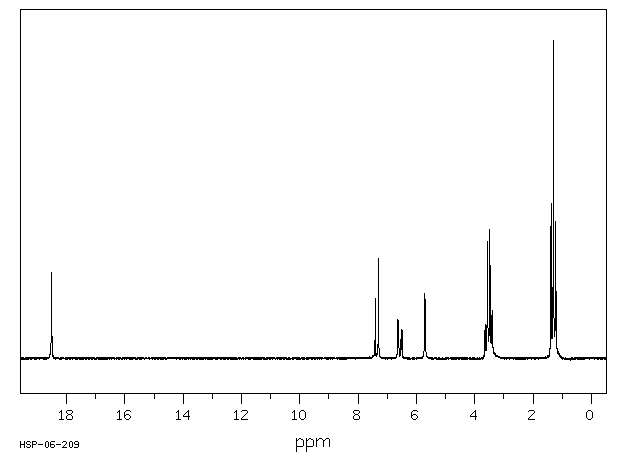
氢谱1HNMR
-
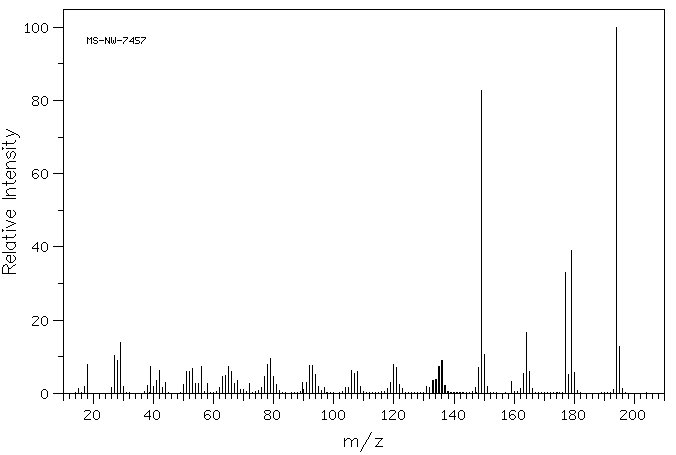
质谱MS
-
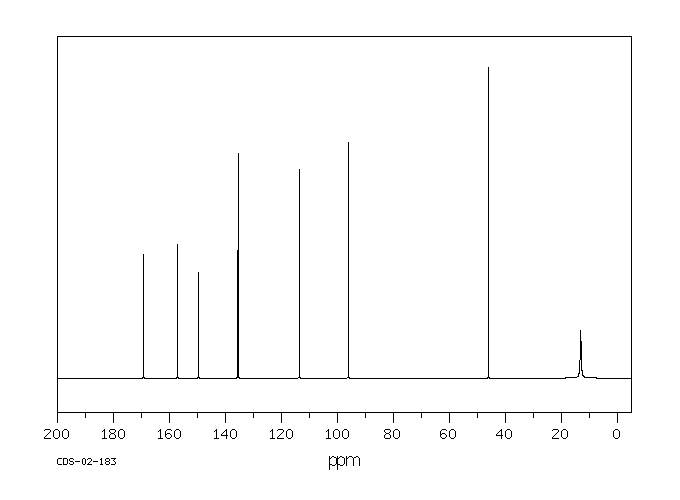
碳谱13CNMR
-
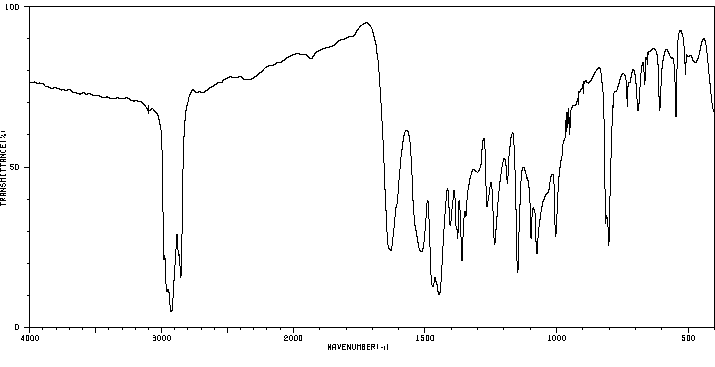
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(乙腈)二氯镍(II)
(R)-(-)-α-甲基组胺二氢溴化物
(N-(2-甲基丙-2-烯-1-基)乙烷-1,2-二胺)
(4-(苄氧基)-2-(哌啶-1-基)吡啶咪丁-5-基)硼酸
(11-巯基十一烷基)-,,-三甲基溴化铵
鼠立死
鹿花菌素
鲸蜡醇硫酸酯DEA盐
鲸蜡硬脂基二甲基氯化铵
鲸蜡基胺氢氟酸盐
鲸蜡基二甲胺盐酸盐
高苯丙氨醇
高箱鲀毒素
高氯酸5-(二甲氨基)-1-({(E)-[4-(二甲氨基)苯基]甲亚基}氨基)-2-甲基吡啶正离子
高氯酸2-氯-1-({(E)-[4-(二甲氨基)苯基]甲亚基}氨基)-6-甲基吡啶正离子
高氯酸2-(丙烯酰基氧基)-N,N,N-三甲基乙铵
马诺地尔
马来酸氢十八烷酯
马来酸噻吗洛尔EP杂质C
马来酸噻吗洛尔
马来酸倍他司汀
顺式环己烷-1,3-二胺盐酸盐
顺式氯化锆二乙腈
顺式吡咯烷-3,4-二醇盐酸盐
顺式双(3-甲氧基丙腈)二氯铂(II)
顺式3,4-二氟吡咯烷盐酸盐
顺式1-甲基环丙烷1,2-二腈
顺式-二氯-反式-二乙酸-氨-环己胺合铂
顺式-二抗坏血酸(外消旋-1,2-二氨基环己烷)铂(II)水合物
顺式-N,2-二甲基环己胺
顺式-4-甲氧基-环己胺盐酸盐
顺式-4-环己烯-1.2-二胺
顺式-4-氨基-2,2,2-三氟乙酸环己酯
顺式-3-氨基环丁烷甲腈盐酸盐
顺式-2-羟基甲基-1-甲基-1-环己胺
顺式-2-甲基环己胺
顺式-2-(苯基氨基)环己醇
顺式-2-(苯基氨基)环己醇
顺式-2-(氨基甲基)-1-苯基环丙烷羧酸盐酸盐
顺式-1,3-二氨基环戊烷
顺式-1,2-环戊烷二胺二盐酸盐
顺式-1,2-环戊烷二胺
顺式-1,2-环丁腈
顺式-1,2-双氨甲基环己烷
顺式--N,N'-二甲基-1,2-环己二胺
顺式-(R,S)-1,2-二氨基环己烷铂硫酸盐
顺式-(2-氨基-环戊基)-甲醇
顺-2-戊烯腈
顺-1,3-环己烷二胺
顺-1,3-双(氨甲基)环己烷