3-(5-methylthiophen-2-yl)acrylic acid | 14770-88-8
中文名称
——
中文别名
——
英文名称
3-(5-methylthiophen-2-yl)acrylic acid
英文别名
3-(5-Methyl-2-thienyl)acrylic acid;3-(5-methylthiophen-2-yl)prop-2-enoic acid
CAS
14770-88-8
化学式
C8H8O2S
mdl
——
分子量
168.216
InChiKey
MHOAQQYXASUJOT-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
计算性质
-
辛醇/水分配系数(LogP):2
-
重原子数:11
-
可旋转键数:2
-
环数:1.0
-
sp3杂化的碳原子比例:0.12
-
拓扑面积:65.5
-
氢给体数:1
-
氢受体数:3
安全信息
-
危险性防范说明:P261,P264,P270,P271,P280,P301+P312,P302+P352,P304+P340,P305+P351+P338,P330,P332+P313,P337+P313,P362,P403+P233,P405,P501
-
危险性描述:H302,H315,H319,H335
-
储存条件:存储条件:2-8°C,避光保存,干燥密封。
SDS
上下游信息
-
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 3-(5-Methylthiophen-2-yl)prop-2-enoyl azide 59207-22-6 C8H7N3OS 193.229
反应信息
-
作为反应物:描述:3-(5-methylthiophen-2-yl)acrylic acid 在 氯化亚砜 、 sodium azide 、 二苯醚 、 N,N-二甲基甲酰胺 作用下, 以 1,4-二氧六环 、 氯仿 、 水 为溶剂, 反应 4.5h, 生成 2-甲基-噻吩[3,2-C]吡啶-4(5H)-酮参考文献:名称:[EN] A METHOD FOR THE SITE-SPECIFIC ENZYMATIC LABELLING OF NUCLEIC ACIDS IN VITRO BY INCORPORATION OF UNNATURAL NUCLEOTIDES
[FR] PROCÉDÉ D'ÉTIQUETAGE ENZYMATIQUE SPÉCIFIQUE DE SITE D'ACIDES NUCLÉIQUES IN VITRO PAR INCORPORATION DE NUCLÉOTIDES NON NATURELS摘要:本文提供了一些不自然核苷酸的类似物,主要带有疏水性核碱基类似物,这些类似物在DNA聚合酶介导的DNA复制或RNA聚合酶介导的RNA转录过程中形成不自然碱基对。通过这种方式,可以将这些不自然碱基以特定位置的方式引入寡核苷酸(单链或双链DNA或RNA)中,从而可以实现特定位置的切割,或者可以提供一个反应性连接物,通过与主要氨基团反应或与不自然碱基连接物的炔基团进行点击化学反应,从而实现与携带试剂的载体的官能化。公开号:WO2015021432A1 -
作为产物:描述:ethyl 3-(5-methylthiophen-2-yl)acrylate 在 sodium hydroxide 作用下, 以 甲醇 、 水 为溶剂, 反应 0.5h, 生成 3-(5-methylthiophen-2-yl)acrylic acid参考文献:名称:In vivo phenotypic drug discovery: applying a behavioral assay to the discovery and optimization of novel antipsychotic agents摘要:一种基于老鼠的检测方法(SmartCube)被用于一种新型抗精神病药物的筛选和首选优化。DOI:10.1039/c6md00128a
文献信息
-
N-substituted nonaryl-heterocyclic NMDA/NR2B antagonists
-
[EN] HETEROARYL COMPOUNDS AND METHODS OF USE THEREOF<br/>[FR] COMPOSÉS HÉTÉROARYLES ET PROCÉDÉ D'UTILISATION CORRESPONDANT申请人:SUNOVION PHARMACEUTICALS INC公开号:WO2013119895A1公开(公告)日:2013-08-15Provided herein are thiophene compounds, methods of their synthesis, pharmaceutical compositions comprising the compounds, and methods of their use. The compounds provided herein are useful for the treatment, prevention, and/or management of various neurological disorders, including but not limited to, psychosis and schizophrenia.本文提供了噻吩化合物,其合成方法,包含这些化合物的药物组合物,以及它们的使用方法。本文提供的化合物可用于治疗、预防和/或管理各种神经系统疾病,包括但不限于精神病和精神分裂症。
-
Synthesis and Sar Study of Diarylpentanoid Analogues as New Anti-Inflammatory Agents作者:Sze Leong、Siti Faudzi、Faridah Abas、Mohd Aluwi、Kamal Rullah、Lam Wai、Mohd Bahari、Syahida Ahmad、Chau Tham、Khozirah Shaari、Nordin LajisDOI:10.3390/molecules191016058日期:——A series of ninety-seven diarylpentanoid derivatives were synthesized and evaluated for their anti-inflammatory activity through NO suppression assay using interferone gamma (IFN-γ)/lipopolysaccharide (LPS)-stimulated RAW264.7 macrophages. Twelve compounds (9, 25, 28, 43, 63, 64, 81, 83, 84, 86, 88 and 97) exhibited greater or similar NO inhibitory activity in comparison with curcumin (14.7 ± 0.2 µM), notably compounds 88 and 97, which demonstrated the most significant NO suppression activity with IC50 values of 4.9 ± 0.3 µM and 9.6 ± 0.5 µM, respectively. A structure–activity relationship (SAR) study revealed that the presence of a hydroxyl group in both aromatic rings is critical for bioactivity of these molecules. With the exception of the polyphenolic derivatives, low electron density in ring-A and high electron density in ring-B are important for enhancing NO inhibition. Meanwhile, pharmacophore mapping showed that hydroxyl substituents at both meta- and para-positions of ring-B could be the marker for highly active diarylpentanoid derivatives.合成了97系列二苯基戊烷类化合物,并通过使用γ干扰素(IFN-γ)/脂多糖(LPS)刺激的RAW264.7巨噬细胞进行NO抑制试验评估其抗炎活性。12个化合物(9, 25, 28, 43, 63, 64, 81, 83, 84, 86, 88和97)与姜黄素(14.7 ± 0.2 µM)相比显示出更大或相似的NO抑制活性,特别是化合物88和97,它们表现出最显著的NO抑制活性,IC50值分别为4.9 ± 0.3 µM和9.6 ± 0.5 µM。结构-活性关系(SAR)研究表明,两个芳香环上羟基的存在对这些分子的生物活性至关重要。除了多酚类衍生物外,A环上低电子密度和B环上高电子密度对提高NO抑制作用很重要。同时,药效团映射表明,B环上间位和对位上的羟基取代可能是高活性二苯基戊烷类化合物的标记。
-
Ramoplanin derivatives possessing antibacterial activity申请人:Raju G. Bore公开号:US20060211603A1公开(公告)日:2006-09-21Novel ramoplanin derivatives are disclosed. These ramoplanin derivatives exhibit antibacterial activity. As the compounds of the subject invention exhibit potent activities against gram positive bacteria, they are useful antimicrobial agents. Methods of synthesis and of use of the compounds are also disclosed.
-
[EN] PROPENAMIDE THIOPHENE DERIVATIVES AS FLAVIVIRUS INHIBITORS AND THEIR USE<br/>[FR] DÉRIVÉS DE THIOPHÈNE PROPÉNAMIDE UTILISÉS COMME INHIBITEURS DE FLAVIVIRUS ET LEUR UTILISATION申请人:UNIV D'AIX-MARSEILLE公开号:WO2017102014A1公开(公告)日:2017-06-22The present invention deals with new flavivirus inhibitors, compositions comprising said inhibitors and methods for the treatment of disorders related to a viral infection, such as a disease due to a flavivirus infection, comprising administering said inhibitors.
表征谱图
-
氢谱1HNMR
-
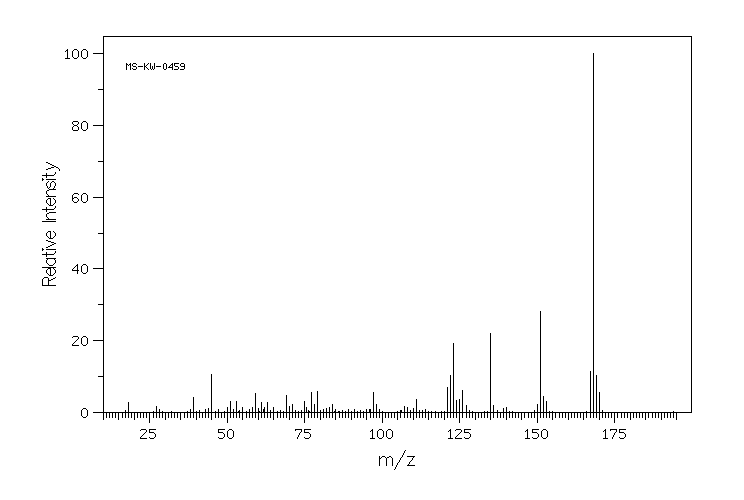
质谱MS
-
碳谱13CNMR
-
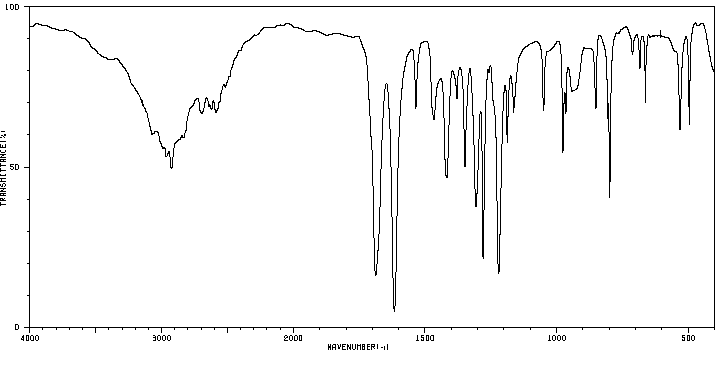
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
阿罗洛尔
阿替卡因
阿克兰酯
锡烷,(5-己基-2-噻吩基)三甲基-
邻氨基噻吩(2盐酸)
辛基5-(1,3-二氧戊环-2-基)-2-噻吩羧酸酯
辛基4,6-二溴噻吩并[3,4-b]噻吩-2-羧酸酯
辛基2-甲基异巴豆酸酯
血管紧张素IIAT2受体激动剂
葡聚糖凝胶LH-20
苯螨噻
苯并[c]噻吩-1-羧酸,5-溴-4,5,6,7-四氢-3-(甲硫基)-4-羰基-,乙基酯
苯并[b]噻吩-2-胺
苯并[b]噻吩-2-胺
苯基-[5-(4,4,5,5-四甲基-[1,3,2]二氧杂硼烷-2-基)-噻吩-2-基亚甲基]-胺
苯基-(5-氯噻吩-2-基)甲醇
苯乙酸,-α--[(1-羰基-2-丙烯-1-基)氨基]-
苯乙酰胺,3,5-二氨基-a-羟基-2,4,6-三碘-
苯乙脒,2,6-二氯-a-羟基-
腈氨噻唑
聚(3-丁基噻吩-2,5-二基),REGIOREGULAR
硝呋肼
硅烷,(3-己基-2,5-噻吩二基)二[三甲基-
硅噻菌胺
盐酸阿罗洛尔
盐酸阿罗洛尔
盐酸多佐胺
甲酮,[5-(1-环己烯-1-基)-4-(2-噻嗯基)-1H-吡咯-3-基]-2-噻嗯基-
甲基5-甲酰基-4-甲基-2-噻吩羧酸酯
甲基5-乙氧基-3-羟基-2-噻吩羧酸酯
甲基5-乙基-3-肼基-2-噻吩羧酸酯
甲基5-(氯甲酰基)-2-噻吩羧酸酯
甲基5-(氯乙酰基)-2-噻吩羧酸酯
甲基5-(氨基甲基)噻吩-2-羧酸酯
甲基5-(4-甲氧基苯基)-2-噻吩羧酸酯
甲基5-(4-甲基苯基)-2-噻吩羧酸酯
甲基5-(1,3-二氧戊环-2-基)-2-噻吩羧酸酯
甲基4-硝基-2-噻吩羧酸酯
甲基4-氰基-5-(4,6-二氨基吡啶-2-基)偶氮-3-甲基噻吩-2-羧酸酯
甲基4-氨基-5-(甲硫基)-2-噻吩羧酸酯
甲基4-{[(2E)-2-(4-氰基苯亚甲基)肼基]磺酰}噻吩-3-羧酸酯
甲基4-(氯甲酰基)-3-噻吩羧酸酯
甲基4-(氨基磺酰基氨基)-3-噻吩羧酸酯
甲基3-甲酰氨基-4-甲基-2-噻吩羧酸酯
甲基3-氨基-5-异丙基-2-噻吩羧酸酯
甲基3-氨基-5-(4-溴苯基)-2-噻吩羧酸酯
甲基3-氨基-4-苯基-5-(三氟甲基)-2-噻吩羧酸酯
甲基3-氨基-4-氰基-5-甲基-2-噻吩羧酸酯
甲基3-氨基-4-丙基-2-噻吩羧酸酯
甲基3-[[(4-甲氧基苯基)亚甲基氨基]氨基磺酰基]噻吩-2-羧酸酯