1-硫代色原酮 | 491-39-4
中文名称
1-硫代色原酮
中文别名
硫色酮;1,4-硫萘酮
英文名称
thiochromen-4-one
英文别名
4H-thiochromen-4-one;thiochromone;1-thiochromone;4H-1-Benzothiopyran-4-one
CAS
491-39-4
化学式
C9H6OS
mdl
——
分子量
162.212
InChiKey
DHSYDTXGFJPXKA-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
计算性质
-
辛醇/水分配系数(LogP):2.1
-
重原子数:11
-
可旋转键数:0
-
环数:2.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:42.4
-
氢给体数:0
-
氢受体数:2
SDS
上下游信息
反应信息
-
作为反应物:参考文献:名称:2,3-二氢-4H-1-苯并噻喃-4-酮和4H-1-苯并吡喃-4-酮的二甲基二环氧乙烷氧化中的化学和非对映选择性。4H-1-苯并噻喃-4-酮1-氧化物的反常活性。摘要:通过二甲基二环氧乙烷(DMD)氧化1-硫代发色酮1-3产生相应的亚砜4-6或砜7-9;它们的相对量取决于所用氧化剂的量。由于在DMD攻击期间小的空间差异,在2-取代的1-硫代发色酮2和3的磺氧化中观察到低的非对映选择性。在1-硫代色酮10-12的DMD氧化中发现了一种不寻常的反应模式,其中亚砜13-15对亲电子氧化剂的反应性高于相应的硫化物。所观察到的异常现象可以通过砜形成的过渡结构(TS)的环空稳定来解释,该结构通过亚砜中的有利构象效应而得以促进。DOI:10.1021/jo001469f
-
作为产物:参考文献:名称:Rh催化的芳基氯化物与噻吩色酮的共轭加成:获得手性硫代黄酮的高对映选择性的途径。摘要:通过使用Rh(COD)Cl 2 /(R)-3,4,5-MeO-MeOBIPHEP催化剂将芳基锌试剂共轭添加到硫代色酮中,开发了一种高效的手性硫代黄酮酮的不对称合成方法。此方法克服了催化剂中毒和底物惰性的问题,并以良好的收率(最高91%的收率)和优异的ee值(最高ee的97%)提供了一系列手性硫代黄烷酮(2-芳基硫代色烷-4-酮)。既定的不对称合成为进一步的药物研究铺平了道路。DOI:10.1021/acs.orglett.6b02453
-
作为试剂:参考文献:名称:硫代色酮在芳族溶剂中的光脱二甲基化摘要:硫代色酮1a在几种电离势相差很大的芳族溶剂中的光解导致形成所有四种可能的环丁烷光二聚体的混合物。与类似砜的光化学行为形成鲜明对比的是,未发现{2 + 2}或取代基的光加成反应的证据。DOI:10.1016/s0040-4039(01)90269-2
文献信息
-
Cu-Catalyzed Conjugate Addition of Grignard Reagents to Thiochromones: An Enantioselective Pathway for Accessing 2-Alkylthiochromanones作者:Qingxiong Yang、Jun Wang、Shihui Luo、Ling MengDOI:10.1055/s-0037-1610225日期:2018.9The enantioselective incorporation of alkyl groups in thiochromones was realized for the first time by a Cu/(R,S)-PPF-P t Bu2-catalyzed conjugate addition of Grignard reagents to thiochromones. With this method, a series of 2-methylthiochromanones were obtained in good yields (up to 96% yield) with moderate-to-good ee values (up to 87% ee). The established method expedites the synthesis of a large
-
Synthesis of Enones and Enals via Dehydrogenation of Saturated Ketones and Aldehydes作者:Gao-Fei Pan、Xue-Qing Zhu、Rui-Li Guo、Ya-Ru Gao、Yong-Qiang WangDOI:10.1002/adsc.201801058日期:2018.12.21substrate scope including various linear or cyclic saturated ketones and aldehydes. The protocol is ligand‐free, and molecular oxygen is used as the sole clean oxidant in the reaction. Due to mild reaction conditions, good functional group compatibility, and versatile utilities of enones and enals, the method can be applied in the late‐stage synthesis of natural products, pharmaceuticals and fine chemicals
-
Synthesis of benzothiazonine by rhodium-catalyzed denitrogenative transannulation of 1-sulfonyl-1,2,3-triazole and thiochromone作者:Saygbechi T. Jablasone、Zihang Ye、Shengguo Duan、Ze-Feng Xu、Chuan-Ying LiDOI:10.1039/d1ob00419k日期:——
A facile synthesis of functionalized benzothiazonine was achieved by rhodium-catalyzed denitrogenative annulation of easily available 1-sulfonyl-1,2,3-triazole and thiochromone.
-
Development of Conjugate Addition of Lithium Dialkylcuprates to Thiochromones: Synthesis of 2-Alkylthiochroman-4-ones and Additional Synthetic Applications作者:Shekinah Bass、Dynasty Parker、Tania Bellinger、Aireal Eaton、Angelica Dibble、Kaata Koroma、Sylvia Sekyi、David Pollard、Fenghai GuoDOI:10.3390/molecules23071728日期:——Lithium dialkylcuprates undergo conjugate addition to thiochromones to afford 2-alkylthiochroman-4-ones in good yields. This approach provide an efficient and general synthetic approach to privileged sulfur-containing structural motifs and valuable precursors for many pharmaceuticals, starting from common substrates-thiochromones. Good yields of 2-alkyl-substituted thiochroman-4-ones are attained with
-
Cu(I)-Catalyzed Enantioselective Alkynylation of Thiochromones作者:Ling Meng、Ka Yan Ngai、Xiaoyong Chang、Zhenyang Lin、Jun WangDOI:10.1021/acs.orglett.0c00005日期:2020.2.7A highly efficient asymmetric synthesis of chiral thiochromanones is developed via Cu(I)/phosphoramidite catalyzed asymmetric alkynylation of thiochromones under mild reaction conditions. The catalyst system is tolerant of various thiochromone precursors and terminal alkynes. The established asymmetric transformation provides different enatiomeric-enriched thiochromanones with more molecular complexity
表征谱图
-
氢谱1HNMR
-
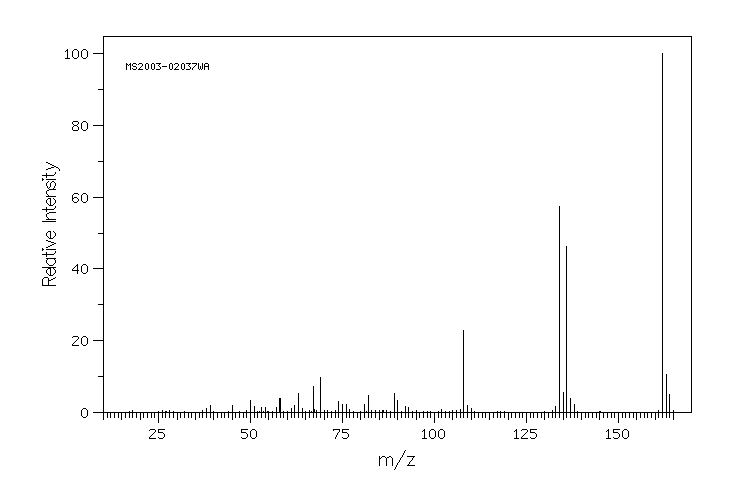
质谱MS
-
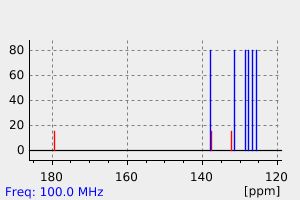
碳谱13CNMR
-
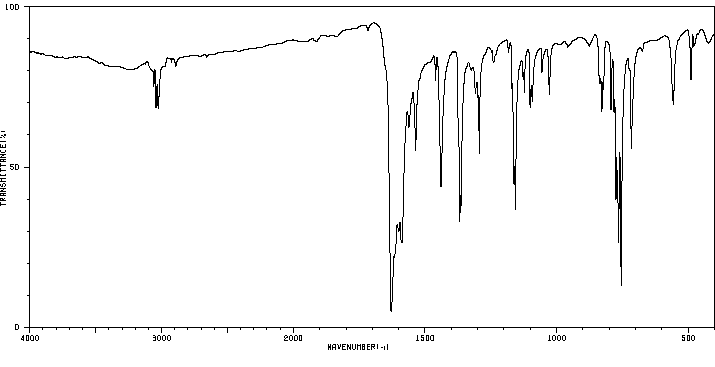
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
贝恩酮盐酸盐
苯并噻喃并[4,3-b]吲哚
苯并[e][1]苯并噻喃并[4,3-b]吲哚
苯并[c]噻吨-7-酮
苯并[a]噻吨-12-酮
硫坎酮
硫代色烯-2-酮
海蒽酮甲磺酸盐
海蒽酮N-甲基氨基甲酸酯
海恩酮
来多蒽琼
异丙基硫代呫吨酮
噻吨酮-3-甲酰胺
[[(9-氧代-9H-噻吨-2-基)甲基]硫代]乙酸
N-[2-(二甲氨基)乙基]-9-羰基-9H-硫代占吨-4-甲酰胺
N,N-二甲基-N'-4H-硫代色烯-4-基酰亚胺基甲酰胺
N'-[4-(羟基甲基)-9-氧代-9H-噻吨-1-基]-N,N-二乙基乙烷-1,2-二胺N-氧化物
9-氧代噻吩-4-羧酸乙酯
9-氧代噻吨-1-羧酸甲酯
9-氧代-9h-硫代氧杂蒽-2-羧酸
9-氧代-9h-硫代氧杂蒽-2-羧酸
9-氧代-9H-硫代氧杂蒽-1-羧酸
9-氧代-9H-噻吨-3-甲腈
9-氧代-9H-噻吨-2-羧酸乙酯
9-氧代-3-(苯基磺酰基)-9H-噻吨-1-羧酸乙酯
9-噻吨酮
8H-苯并噻喃并[7,8-D][1,3]噻唑
8H-苯并噻喃并[6,7-D][1,3]噻唑
8-甲基-4H-硫色烯-4-酮
8-氯-N,N-二乙基-5-甲基-2H-[1]苯并噻喃并[4,3,2-cd]吲唑-2-乙胺N-氧化物
8-氯-5-甲基-N,N-二乙基-2H-[1]苯并噻喃并[4,3,2-cd]吲唑-2-乙烷-1-胺
8-氯-5-(羟基甲基)-N,N-二乙基-2H-[1]苯并噻喃并[4,3,2-cd]吲唑-2-乙烷-1-胺N-氧化物
7H-苯并噻喃并[6,5-d][1,3]噻唑
7-甲氧基-2-甲基-4H-硫代色烯-4-酮
7-甲基-9-氧代噻吨-3-羧酸乙酯
7-氨基-1-[[2-(二乙胺)乙基]氨基]-4-甲基-L-9H-噻吨-9-酮
6H-苯并噻喃并[7,6-D][1,3]噻唑
6-甲氧基-2-甲基-4H-硫代色烯-4-酮
6-甲基-4H-硫代色烯-4-酮
6-溴-4H-硫代色素-4-酮
6-氯-4H-硫色烯-4-酮
6-氯-1-({2-[乙基(2-羟基乙基)氨基]乙基}氨基)-4-(羟甲基)-9H-硫代占吨-9-酮
6-氟-4H-硫代色烯-4-酮
6-氟-2-(三氟甲基)-9H-噻吨-9-酮
6-(2-二乙基氨基乙胺)-1,2,3,4-四氢苯并[a]噻吨-12-酮
5-[(2-氨基乙基)氨基]-2-[2-(二乙基氨基)乙基]-2H-苯并噻喃并[4,3,2-Cd]吲唑-8-醇三盐酸盐
5-(2-二乙基氨基乙胺)-1,2,3,4-四氢苯并[c]噻吨-7-酮
4H-1-苯并噻喃-4-酮,3-[(3-硝基苯基)甲基]-2-苯基-,1,1-二氧化
4H-1-苯并噻喃-4-酮,3-[(2-溴苯基)甲基]-2-苯基-,1,1-二氧化
4H-1-苯并噻喃-4-酮 1,1-二氧化物