4,5-二溴噻吩-2-磺酰氯 | 81606-31-7
中文名称
4,5-二溴噻吩-2-磺酰氯
中文别名
4,5-二溴噻酚-2-磺酰氯
英文名称
4,5-dibromo-thiophene-2-sulfonyl chloride
英文别名
4,5-dibromothiophene-2-sulphonyl chloride;4,5-dibromo-2-thiophenesulfonyl chloride;2,3-dibromothiophene-5-sulfonyl chloride;4,5-Dibrom-thiophen-2-sulfonylchlorid;4,5-Dibromothiophene-2-sulfonyl chloride
CAS
81606-31-7
化学式
C4HBr2ClO2S2
mdl
MFCD00041439
分子量
340.444
InChiKey
WJYGHWXWQSCONR-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:71-75 °C
计算性质
-
辛醇/水分配系数(LogP):3.7
-
重原子数:11
-
可旋转键数:1
-
环数:1.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:70.8
-
氢给体数:0
-
氢受体数:3
安全信息
-
危险等级:8
-
危险品标志:C
-
安全说明:S26,S36/37/39,S45
-
危险类别码:R34
-
海关编码:2934999090
-
包装等级:II
-
危险类别:8
-
危险品运输编号:3261
SDS
| Name: | 4 5-Dibromothiophene-2-sulfonyl chloride Material Safety Data Sheet |
| Synonym: | |
| CAS: | 81606-31-7 |
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 81606-31-7 | 4,5-Dibromothiophene-2-sulfonyl chlori | unlisted |
Risk Phrases: 34
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Causes burns.Moisture sensitive.
Potential Health Effects
Eye:
Causes eye burns.
Skin:
Causes skin burns.
Ingestion:
Causes gastrointestinal tract burns.
Inhalation:
Causes chemical burns to the respiratory tract. Inhalation may be fatal as a result of spasm, inflammation, edema of the larynx and bronchi, chemical pneumonitis and pulmonary edema. May cause burning sensation, coughing, wheezing, laryngitis, shortness of breath, headache, nausea, and vomiting.
Chronic:
Not available.
Section 4 - FIRST AID MEASURES
Eyes: Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid immediately.
Skin:
Get medical aid immediately. Immediately flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:
Get medical aid immediately. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Get medical aid immediately. Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or chemical foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container.
Section 7 - HANDLING and STORAGE
Handling:
Do not breathe dust, vapor, mist, or gas. Do not get in eyes, on skin, or on clothing. Use only in a chemical fume hood.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Corrosives area.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 81606-31-7: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Solid
Color: Not available.
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 75 - 77 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
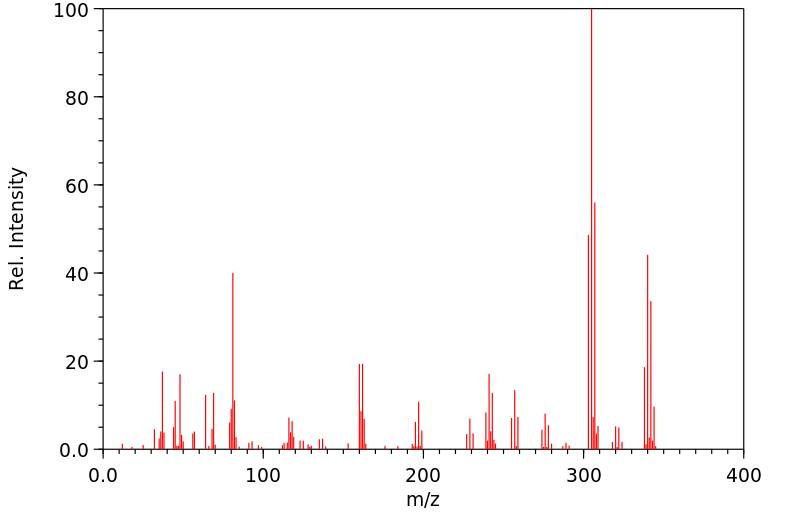
Molecular Formula: C4HBr2ClO2S2
Molecular Weight: 340.44
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Incompatible materials, exposure to moist air or water.
Incompatibilities with Other Materials:
Strong oxidizing agents, strong acids.
Hazardous Decomposition Products:
Hydrogen chloride, sulfur dioxide, carbon monoxide, carbon dioxide, hydrogen bromide, sulfuric acid.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 81606-31-7 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
4,5-Dibromothiophene-2-sulfonyl chloride - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Other No information available.
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Shipping Name: CORROSIVE SOLID, N.O.S.*
Hazard Class: 8
UN Number: 1759
Packing Group: III
IMO
Shipping Name: CORROSIVE SOLID, N.O.S.
Hazard Class: 8
UN Number: 1759
Packing Group: III
RID/ADR
No information available.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: C
Risk Phrases:
R 34 Causes burns.
Safety Phrases:
S 26 In case of contact with eyes, rinse immediately
with plenty of water and seek medical advice.
S 36/37/39 Wear suitable protective clothing, gloves
and eye/face protection.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 81606-31-7: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 81606-31-7 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 81606-31-7 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
下游产品
中文名称 英文名称 CAS号 化学式 分子量 4,5-二溴噻吩-2-磺酰胺 2,3-dibromo-5-thiophenesulfonamide 77893-69-7 C4H3Br2NO2S2 321.013
反应信息
-
作为反应物:描述:4,5-二溴噻吩-2-磺酰氯 以 甲苯 为溶剂, 生成 C4HBr2O2S2参考文献:名称:噻吩-2-磺酰基自由基的结构和构象研究摘要:描述了各种光化学产生的噻吩-2-磺酰基的esr光谱。它们的自旋分布是σ自由基的典型特征。在所有可及的温度下,位置3处没有取代基的自由基均表现出相对较快的绕C–S键旋转,而3-溴取代的自由基则表现出显着的构象偏好,这是根据π型共轭结构来解释的。这些发现也被对具有SO 2和杂芳族部分的不同相对排列的未取代的噻吩-2-磺酰基基团进行的INDO计算结果所证实。DOI:10.1039/p29860001179
-
作为产物:描述:参考文献:名称:噻吩-2-磺酰基自由基的结构和构象研究摘要:描述了各种光化学产生的噻吩-2-磺酰基的esr光谱。它们的自旋分布是σ自由基的典型特征。在所有可及的温度下,位置3处没有取代基的自由基均表现出相对较快的绕C–S键旋转,而3-溴取代的自由基则表现出显着的构象偏好,这是根据π型共轭结构来解释的。这些发现也被对具有SO 2和杂芳族部分的不同相对排列的未取代的噻吩-2-磺酰基基团进行的INDO计算结果所证实。DOI:10.1039/p29860001179
文献信息
-
FKBP BINDING COMPOSITION AND PHARMACEUTICAL USE THEREOF申请人:KOSLEY Raymond W.公开号:US20080139556A1公开(公告)日:2008-06-12A composition for binding FKBP proteins is disclosed, along with a method of treating conditions associated with neuronal degeneration, wherein said composition comprises a compound of formula I, wherein, R, R 1 , R 2 , R 3 and X are as defined herein.揭示了一种用于结合FKBP蛋白的组合物,以及一种治疗与神经退行性疾病相关的条件的方法,其中所述组合物包括式I的化合物, 其中,R、R1、R2、R3和X如本文所定义。
-
Small-Molecule Inhibitors That Target Protein-Protein Interactions in the RAD51 Family of Recombinases作者:Duncan E. Scott、Anthony G. Coyne、Ashok Venkitaraman、Tom L. Blundell、Chris Abell、Marko HyvönenDOI:10.1002/cmdc.201402428日期:2015.2The development of small molecules that inhibit protein–protein interactions continues to be a challenge in chemical biology and drug discovery. Herein we report the development of indole‐based fragments that bind in a shallow surface pocket of a humanised surrogate of RAD51. RAD51 is an ATP‐dependent recombinase that plays a key role in the repair of double‐strand DNA breaks. It both self‐associates抑制蛋白质与蛋白质相互作用的小分子的发展仍然是化学生物学和药物发现中的挑战。在这里,我们报道了结合吲哚基片段的发展,该片段结合在RAD51人性化替代物的浅表囊中。RAD51是一种依赖于ATP的重组酶,在双链DNA断裂的修复中起关键作用。它既可以自缔合,与DNA形成细丝结构,又可以通过常见的“ FxxA”四肽基序与BRCA2蛋白相互作用。我们精心设计了先前确定的靶向FxxA序列位点的片段,并开发了比初始片段强约500倍的小分子抑制剂。铅化合物与BRCA2衍生的Ac-FHTA-NH 2竞争肽和RAD51的自缔合肽,但它们对ATP结合没有影响。这项研究是首次报道针对这一具有挑战性的目标的小分子量片段的研究。
-
BENZIMIDAZOLE CANNABINOID AGONISTS BEARING A SUBSTITUTED HETEROCYCLIC GROUP申请人:JANSSEN PHARMACEUTICA NV公开号:US20130324529A1公开(公告)日:2013-12-05The present invention is related to novel benzimidazole compounds of formula (I) having cannabinoid receptor agonistic properties, pharmaceutical compositions comprising these compounds, chemical processes for preparing these compounds and their use in the treatment of diseases linked to the mediation of the cannabinoid receptors in animals, in particular humans.
-
Tyrosine alkoxyguanidines as integrin inhibitors申请人:3-Dimensional Pharmaceuticals, Inc.公开号:US06344484B1公开(公告)日:2002-02-05The present invention relates to novel tyrosine alkoxyguanidine compounds that are inhibitors of alpha V (&agr;v) integrins, for example &agr;v&bgr;3 and &agr;v&bgr;5 integrins, their pharmaceutically acceptable salts, and pharmaceutical compositions thereof. The compounds may be used in the treatment of pathological conditions mediated by &agr;v&bgr;3 and &agr;v&bgr;5 integrins, including conditions such as tumor growth, metastasis, restenosis, osteoporosis, inflammation, macular degeneration, diabetic retinopathy, and rheumatoid arthritis. The compounds have the general formula: where R1, R2, R3, R4, R5, R6, R7, R8, R9, R10, R11, m and n are defined herein.
-
[EN] FUSED BICYCLIC COMPOUNDS AND USE THEREOF AS PI3K INHIBITORS<br/>[FR] COMPOSÉS BICYCLIQUES FUSIONNÉS ET UTILISATIONS DE CEUX-CI COMME INHIBITEURS DE P13K申请人:MERCK SERONO SA公开号:WO2009133127A1公开(公告)日:2009-11-05The invention relates to compounds of formula (I), for the regulation of phosphoinositides 3-kinases activity and related diseases.这项发明涉及到式(I)的化合物,用于调节磷脂酰肌醇3-激酶的活性和相关疾病。
表征谱图
-
氢谱1HNMR
-
质谱MS
-
碳谱13CNMR
-
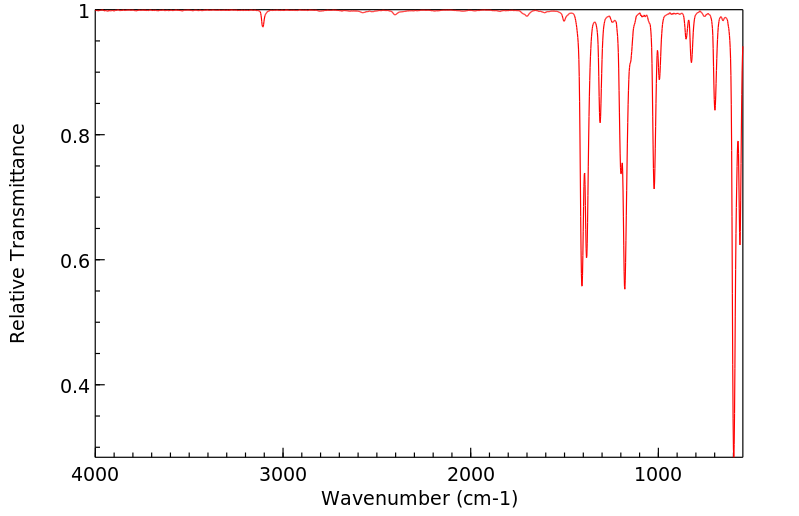
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
阿罗洛尔
阿替卡因
阿克兰酯
锡烷,(5-己基-2-噻吩基)三甲基-
邻氨基噻吩(2盐酸)
辛基5-(1,3-二氧戊环-2-基)-2-噻吩羧酸酯
辛基4,6-二溴噻吩并[3,4-b]噻吩-2-羧酸酯
辛基2-甲基异巴豆酸酯
血管紧张素IIAT2受体激动剂
葡聚糖凝胶LH-20
苯螨噻
苯并[c]噻吩-1-羧酸,5-溴-4,5,6,7-四氢-3-(甲硫基)-4-羰基-,乙基酯
苯并[b]噻吩-2-胺
苯并[b]噻吩-2-胺
苯基-[5-(4,4,5,5-四甲基-[1,3,2]二氧杂硼烷-2-基)-噻吩-2-基亚甲基]-胺
苯基-(5-氯噻吩-2-基)甲醇
苯乙酸,-α--[(1-羰基-2-丙烯-1-基)氨基]-
苯乙酰胺,3,5-二氨基-a-羟基-2,4,6-三碘-
苯乙脒,2,6-二氯-a-羟基-
腈氨噻唑
聚(3-丁基噻吩-2,5-二基),REGIOREGULAR
硝呋肼
硅烷,(3-己基-2,5-噻吩二基)二[三甲基-
硅噻菌胺
盐酸阿罗洛尔
盐酸阿罗洛尔
盐酸多佐胺
甲酮,[5-(1-环己烯-1-基)-4-(2-噻嗯基)-1H-吡咯-3-基]-2-噻嗯基-
甲基5-甲酰基-4-甲基-2-噻吩羧酸酯
甲基5-乙氧基-3-羟基-2-噻吩羧酸酯
甲基5-乙基-3-肼基-2-噻吩羧酸酯
甲基5-(氯甲酰基)-2-噻吩羧酸酯
甲基5-(氯乙酰基)-2-噻吩羧酸酯
甲基5-(氨基甲基)噻吩-2-羧酸酯
甲基5-(4-甲氧基苯基)-2-噻吩羧酸酯
甲基5-(4-甲基苯基)-2-噻吩羧酸酯
甲基5-(1,3-二氧戊环-2-基)-2-噻吩羧酸酯
甲基4-硝基-2-噻吩羧酸酯
甲基4-氰基-5-(4,6-二氨基吡啶-2-基)偶氮-3-甲基噻吩-2-羧酸酯
甲基4-氨基-5-(甲硫基)-2-噻吩羧酸酯
甲基4-{[(2E)-2-(4-氰基苯亚甲基)肼基]磺酰}噻吩-3-羧酸酯
甲基4-(氯甲酰基)-3-噻吩羧酸酯
甲基4-(氨基磺酰基氨基)-3-噻吩羧酸酯
甲基3-甲酰氨基-4-甲基-2-噻吩羧酸酯
甲基3-氨基-5-异丙基-2-噻吩羧酸酯
甲基3-氨基-5-(4-溴苯基)-2-噻吩羧酸酯
甲基3-氨基-4-苯基-5-(三氟甲基)-2-噻吩羧酸酯
甲基3-氨基-4-氰基-5-甲基-2-噻吩羧酸酯
甲基3-氨基-4-丙基-2-噻吩羧酸酯
甲基3-[[(4-甲氧基苯基)亚甲基氨基]氨基磺酰基]噻吩-2-羧酸酯