三乙氧基硅烷 | 998-30-1
中文名称
三乙氧基硅烷
中文别名
三乙氧矽烷;三乙氧基矽烷;偶联剂(三乙氧基硅烷)
英文名称
Triethoxysilane
英文别名
(EtO)3SiH;Hydridotriethoxysilane
CAS
998-30-1
化学式
C6H16O3Si
mdl
MFCD00009061
分子量
164.277
InChiKey
QQQSFSZALRVCSZ-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:-170°C
-
沸点:134-135 °C(lit.)
-
密度:0.89 g/mL at 25 °C(lit.)
-
闪点:80 °F
-
溶解度:sol diethyl ether, THF, alkanes, aromatic and chlorinated solvents.
-
稳定性/保质期:
常温常压下稳定,避免与强氧化剂和水分接触。
计算性质
-
辛醇/水分配系数(LogP):0.73
-
重原子数:10
-
可旋转键数:6
-
环数:0.0
-
sp3杂化的碳原子比例:1.0
-
拓扑面积:27.7
-
氢给体数:0
-
氢受体数:3
安全信息
-
TSCA:Yes
-
危险等级:6.1
-
危险品标志:T,C
-
安全说明:S26,S28,S36/37/39,S45
-
危险类别码:R23/24/25,R10,R34
-
WGK Germany:3
-
海关编码:29209090
-
危险品运输编号:UN 3384 6.1/PG 1
-
危险类别:6.1
-
RTECS号:VV6682000
-
包装等级:II
-
储存条件:存储在干燥的惰性气体中,并确保容器密封。应将其存放在阴凉、干燥的地方。
SDS
Triethoxysilane Revision number: 6
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: Triethoxysilane
Revision number: 6
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS
Flammable liquids Category 3
HEALTH HAZARDS
Acute toxicity (Inhalation) Category 1
Skin corrosion/irritation Category 1B
Serious eye damage/eye irritation Category 1
ENVIRONMENTAL HAZARDS Not classified
GHS label elements, including precautionary statements
Pictograms or hazard symbols
Signal word Danger
Hazard statements Flammable liquid and vapour
Fatal if inhaled
Causes severe skin burns and eye damage
Precautionary statements:
[Prevention] Keep away from heat/sparks/open flames/hot surfaces. - No smoking.
Keep container tightly closed.
Use explosion-proof electrical/ventilating/lighting equipment. Take precautionary
measures against ignition by the static discharge and the spark.
Do not breathe dust/fume/gas/mist/vapours/spray.
Use only outdoors or in a well-ventilated area.
Wash hands thoroughly after handling.
Wear respiratory protection.
Wear protective gloves/eye protection/face protection.
[Response] IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for
breathing. Immediately call a POISON CENTER or doctor/physician.
IF SWALLOWED: Rinse mouth. Do NOT induce vomiting.
IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses,
if present and easy to do. Continue rinsing.
IF ON SKIN (or hair): Remove/Take off immediately all contaminated clothing. Rinse
skin with water/shower.
Wash contaminated clothing before reuse.
Immediately call a POISON CENTER or doctor/physician.
Triethoxysilane
Section 2. HAZARDS IDENTIFICATION
[Storage] Store in a well-ventilated place. Keep container tightly closed.
Store locked up.
[Disposal] Dispose of contents/container through a waste management company authorized by
the local government.
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Components: Triethoxysilane
Percent: >97.0%(GC)
CAS Number: 998-30-1
Chemical Formula: C6H16O3Si
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Immediately call a POISON CENTER or doctor/physician.
Skin contact: Remove/Take off immediately all contaminated clothing. Gently wash with plenty of
soap and water. Immediately call a POISON CENTER or doctor/physician.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing.Immediately call a POISON CENTER or
doctor/physician.
Ingestion: Immediately call a POISON CENTER or doctor/physician. Rinse mouth. Do NOT
induce vomiting.
Most important Cough, Sore throat, Redness, Pain
symptoms/effects, acute
and delayed:
Protection of first-aiders: A rescuer should wear personal protective equipment, such as rubber gloves and
air-tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, carbon dioxide.
media:
Unsuitable extinguishing Water (It may scatter and spread fire.)
media:
Specific hazards arising Take care as it may decompose upon combustion or in high temperatures to
from the chemical: generate poisonous fume.
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Keep containers cool by
spraying with water. Eliminate all ignition sources if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, Use extra personal protective equipment (self-contained breathing apparatus). Keep
protective equipment and people away from and upwind of spill/leak. Ensure adequate ventilation. Entry to
emergency procedures: non-involved personnel should be controlled around the leakage area by roping off,
etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Absorb spilled material in dry sand or inert absorbent before recovering it into an
containment and cleaning airtight container. In case of large amount of spillage, contain a spill by bunding.
up: Adhered or collected material should be promptly disposed of, in accordance with
appropriate laws and regulations.
Prevention of secondary Remove all sources of ignition. Fire-extinguishing devices should be prepared in case
hazards: of a fire. Use spark-proof tools and explosion-proof equipment.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Triethoxysilane
Section 7. HANDLING AND STORAGE
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent generation of vapour or mist. Keep away from heat/sparks/open flame/hot
surfaces. -No smoking. Take measures to prevent the build up of electrostatic
charge. Use explosion-proof equipment. Wash hands and face thoroughly after
handling.
Use a closed system if possible. Use a ventilation, local exhaust if vapour or aerosol
will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
May develop pressure. Open carefully.
Conditions for safe storage, including any
incompatibilities
Storage conditions: Keep container tightly closed. Store in a cool, dark and well-ventilated place.
Store under inert gas.
Protect from moisture.
Store locked up.
Store away from incompatible materials such as oxidizing agents.
Moisture-sensitive
Packaging material: Comply with laws.
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Half or full facepiece respirator, self-contained breathing apparatus(SCBA), supplied
air respirator, etc. Use respirators approved under appropriate government standards
and follow local and national regulations.
Hand protection: Impervious gloves.
Eye protection: Safety goggles. A face-shield, if the situation requires.
Skin and body protection: Impervious protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Liquid
Form: Clear
Colorless - Almost colorless
Colour:
Odour: No data available
pH: No data available
Melting point/freezing point: No data available
Boiling point/range: 134°C
Flash point: 29°C
Flammability or explosive
limits:
Lower: 1.2%
Upper: No data available
Vapour density: 5.7
0.90
Relative density:
Solubility(ies):
[Water] No data available
[Other solvents] No data available
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous Disproportionation can occur in the presence of bases to produce pyrophoric silanes
reactions: occasionally.
Conditions to avoid: Spark, Open flame, Static discharge
Incompatible materials: Oxidizing agents, Acids, Bases, Water, Metals
Hazardous decomposition Carbon monoxide, Carbon dioxide, Silicon oxides
products:
Triethoxysilane
Section 11. TOXICOLOGICAL INFORMATION
ihl-mus LC50:500 mg/m3/2H
Acute Toxicity:
ihl-rat LC50:500 mg/m3/2H
ivn-mus LD50:180 mg/kg
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
NTP = No data available
Reproductive toxicity: No data available
VV6682000
RTECS Number:
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobility in soil
Log Pow: No data available
Soil adsorption (Koc): No data available
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to burn in a chemical
incinerator equipped with an afterburner and scrubber system. Observe all federal, state and local regulations when
disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: 6.1: Toxic substance.
Subsidiary risk: 3/8: Flammable liquid/Corrosive.
UN-No: 3489
Proper shipping name: Toxic by inhalation liquid, flammable, corrosive, n.o.s.
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
Triethoxysilane
SECTION 16 - ADDITIONAL INFORMATION
N/A
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: Triethoxysilane
Revision number: 6
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS
Flammable liquids Category 3
HEALTH HAZARDS
Acute toxicity (Inhalation) Category 1
Skin corrosion/irritation Category 1B
Serious eye damage/eye irritation Category 1
ENVIRONMENTAL HAZARDS Not classified
GHS label elements, including precautionary statements
Pictograms or hazard symbols
Signal word Danger
Hazard statements Flammable liquid and vapour
Fatal if inhaled
Causes severe skin burns and eye damage
Precautionary statements:
[Prevention] Keep away from heat/sparks/open flames/hot surfaces. - No smoking.
Keep container tightly closed.
Use explosion-proof electrical/ventilating/lighting equipment. Take precautionary
measures against ignition by the static discharge and the spark.
Do not breathe dust/fume/gas/mist/vapours/spray.
Use only outdoors or in a well-ventilated area.
Wash hands thoroughly after handling.
Wear respiratory protection.
Wear protective gloves/eye protection/face protection.
[Response] IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for
breathing. Immediately call a POISON CENTER or doctor/physician.
IF SWALLOWED: Rinse mouth. Do NOT induce vomiting.
IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses,
if present and easy to do. Continue rinsing.
IF ON SKIN (or hair): Remove/Take off immediately all contaminated clothing. Rinse
skin with water/shower.
Wash contaminated clothing before reuse.
Immediately call a POISON CENTER or doctor/physician.
Triethoxysilane
Section 2. HAZARDS IDENTIFICATION
[Storage] Store in a well-ventilated place. Keep container tightly closed.
Store locked up.
[Disposal] Dispose of contents/container through a waste management company authorized by
the local government.
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Components: Triethoxysilane
Percent: >97.0%(GC)
CAS Number: 998-30-1
Chemical Formula: C6H16O3Si
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Immediately call a POISON CENTER or doctor/physician.
Skin contact: Remove/Take off immediately all contaminated clothing. Gently wash with plenty of
soap and water. Immediately call a POISON CENTER or doctor/physician.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing.Immediately call a POISON CENTER or
doctor/physician.
Ingestion: Immediately call a POISON CENTER or doctor/physician. Rinse mouth. Do NOT
induce vomiting.
Most important Cough, Sore throat, Redness, Pain
symptoms/effects, acute
and delayed:
Protection of first-aiders: A rescuer should wear personal protective equipment, such as rubber gloves and
air-tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, carbon dioxide.
media:
Unsuitable extinguishing Water (It may scatter and spread fire.)
media:
Specific hazards arising Take care as it may decompose upon combustion or in high temperatures to
from the chemical: generate poisonous fume.
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Keep containers cool by
spraying with water. Eliminate all ignition sources if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, Use extra personal protective equipment (self-contained breathing apparatus). Keep
protective equipment and people away from and upwind of spill/leak. Ensure adequate ventilation. Entry to
emergency procedures: non-involved personnel should be controlled around the leakage area by roping off,
etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Absorb spilled material in dry sand or inert absorbent before recovering it into an
containment and cleaning airtight container. In case of large amount of spillage, contain a spill by bunding.
up: Adhered or collected material should be promptly disposed of, in accordance with
appropriate laws and regulations.
Prevention of secondary Remove all sources of ignition. Fire-extinguishing devices should be prepared in case
hazards: of a fire. Use spark-proof tools and explosion-proof equipment.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Triethoxysilane
Section 7. HANDLING AND STORAGE
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent generation of vapour or mist. Keep away from heat/sparks/open flame/hot
surfaces. -No smoking. Take measures to prevent the build up of electrostatic
charge. Use explosion-proof equipment. Wash hands and face thoroughly after
handling.
Use a closed system if possible. Use a ventilation, local exhaust if vapour or aerosol
will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
May develop pressure. Open carefully.
Conditions for safe storage, including any
incompatibilities
Storage conditions: Keep container tightly closed. Store in a cool, dark and well-ventilated place.
Store under inert gas.
Protect from moisture.
Store locked up.
Store away from incompatible materials such as oxidizing agents.
Moisture-sensitive
Packaging material: Comply with laws.
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Half or full facepiece respirator, self-contained breathing apparatus(SCBA), supplied
air respirator, etc. Use respirators approved under appropriate government standards
and follow local and national regulations.
Hand protection: Impervious gloves.
Eye protection: Safety goggles. A face-shield, if the situation requires.
Skin and body protection: Impervious protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Liquid
Form: Clear
Colorless - Almost colorless
Colour:
Odour: No data available
pH: No data available
Melting point/freezing point: No data available
Boiling point/range: 134°C
Flash point: 29°C
Flammability or explosive
limits:
Lower: 1.2%
Upper: No data available
Vapour density: 5.7
0.90
Relative density:
Solubility(ies):
[Water] No data available
[Other solvents] No data available
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous Disproportionation can occur in the presence of bases to produce pyrophoric silanes
reactions: occasionally.
Conditions to avoid: Spark, Open flame, Static discharge
Incompatible materials: Oxidizing agents, Acids, Bases, Water, Metals
Hazardous decomposition Carbon monoxide, Carbon dioxide, Silicon oxides
products:
Triethoxysilane
Section 11. TOXICOLOGICAL INFORMATION
ihl-mus LC50:500 mg/m3/2H
Acute Toxicity:
ihl-rat LC50:500 mg/m3/2H
ivn-mus LD50:180 mg/kg
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
NTP = No data available
Reproductive toxicity: No data available
VV6682000
RTECS Number:
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobility in soil
Log Pow: No data available
Soil adsorption (Koc): No data available
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to burn in a chemical
incinerator equipped with an afterburner and scrubber system. Observe all federal, state and local regulations when
disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: 6.1: Toxic substance.
Subsidiary risk: 3/8: Flammable liquid/Corrosive.
UN-No: 3489
Proper shipping name: Toxic by inhalation liquid, flammable, corrosive, n.o.s.
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
Triethoxysilane
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 硅酸四乙酯 Tetraethoxysilane 78-10-4 C8H20O4Si 208.33 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— triethoxysilanol 27491-84-5 C6H16O4Si 180.276 —— Si(OMe)(OEt)3 18395-48-7 C7H18O4Si 194.303 硅酸四乙酯 Tetraethoxysilane 78-10-4 C8H20O4Si 208.33 二乙氧基硅烷 diethoxysilane 18165-68-9 C4H12O2Si 120.224 —— tributoxysilane 6485-86-5 C12H28O3Si 248.438 —— tetraethylorthosilicate 23241-74-9 C9H22O4Si 222.357 一氟三乙氧基硅烷 triethoxyfluorosilane 358-60-1 C6H15FO3Si 182.267 —— silicic acid triethyl ester-allyl ester 17865-49-5 C9H20O4Si 220.341 三乙氧基氯硅烷 chlorotriethoxysilane 4667-99-6 C6H15ClO3Si 198.722 —— triethoxysilyl formate —— C7H16O5Si 208.287 —— silicic acid triethyl ester-tert-butyl ester 17980-52-8 C10H24O4Si 236.384 - 1
- 2
反应信息
-
作为反应物:描述:参考文献:名称:STUDIES IN SILICO-ORGANIC COMPOUNDS. VI. THE PREPARATION AND PROPERTIES OF POLYETHERS FROM TRICHLOROSILANE1摘要:DOI:10.1021/jo01160a017
-
作为产物:描述:参考文献:名称:热不稳定的金属氢化物配合物如何即使在高温下也能产生高催化活性摘要:尽管它们在醚溶剂中不稳定,但有机钛氢化物催化剂已成功地用于中高温度(110°C)的催化,甚至在存在醇的情况下。通过计算(键离解能(BDE)分析和再生的能量分布)和实验(EPR研究和动力学研究)以及加氢脱氟化氢(HDF)的具体实例表明,尽管长期以来人们一直相信Ti-H的再生使用硅烷从Ti-F键形成的键是endergonic的。Ti-H物质的低浓度对于这些系统的催化稳定性至关重要。催化中的静止状态是Ti-F物种。用于再生的最有希望的硅烷不是具有最强Si-F键的硅烷,而是在Si-F和Si-H BDE中表现出最大差异的硅烷。DOI:10.1002/chem.201601641
-
作为试剂:描述:6-氧代庚酸甲酯 在 6,6'-二甲基-2,2'-联吡啶 、 氯化镍二甲氧基乙烷 、 三乙氧基硅烷 、 lithium fluoride 作用下, 以 二甲基亚砜 为溶剂, 反应 60.0h, 生成 9,9-difluoro-8-(4-methoxyphenyl)-6,6-dimethyl-1-morpholinonon-8-en-1-one参考文献:名称:NiH催化的迁移性脱氟烯烃交叉偶联:用三氟甲基取代的烯烃作为受体烯烃形成宝石-二氟烯烃。摘要:我们报告了两个电子分化的烯烃之间的镍氢催化迁移脱氟偶联。多种未活化的供体烯烃可以以分子内和分子间方式直接与含有缺电子的三氟甲基取代基的受体烯烃键合,形成宝石-二氟烯烃。这种迁移偶联在温和条件下显示出位选择性和化学选择性,并形成了叔碳或季碳中心。DOI:10.1002/anie.201915840
文献信息
-
SILANE COUPLING COMPOUNDS AND MEDICAL AND/OR DENTAL CURABLE COMPOSITIONS COMPRISING THE SAME申请人:KABUSHIKI KAISHA SHOFU公开号:US20190300552A1公开(公告)日:2019-10-03The present invention relate to a novel silane coupling agent and a medical and/or dental curable composition comprising the same. It is an object of the present invention to provide a novel silane coupling agent that imparts high affinity to a radical polymerizable monomer, thereby imparting high mechanical strength, flexibility and durability when used for a medical and/or dental curable composition, and an inorganic filler surface-treated with the novel silane coupling agent and a novel medical and/or dental curable composition. A silane coupling agent including repeating units such as a urethane bond and polyethylene glycol (ether bond) at a specific position is used.
-
Fe and Co Complexes of Rigidly Planar Phosphino-Quinoline-Pyridine Ligands for Catalytic Hydrosilylation and Dehydrogenative Silylation作者:Debashis Basu、Ryan Gilbert-Wilson、Danielle L. Gray、Thomas B. Rauchfuss、Aswini K. DashDOI:10.1021/acs.organomet.8b00416日期:2018.8.27of simple and complex 1-alkenes with a variety of hydrosilanes. Catalysts derived from MesNQpy exhibited low activity. Fe-RPQpy derived catalysts favor hydrosilylation, whereas Co-RPQpy based catalysts favor dehydrogenative silylation. Catalysts derived from CoX2(iPrPQpy) convert hydrosilanes and ethylene to vinylsilanes. Related experiments were conducted on propylene to give propenylsilanes.制备了一种新的刚性平面PNN配体平台的Co和Fe二卤化物配合物,并将其作为烯烃氢化硅烷化的预催化剂。将Thummel的8-溴-2-(吡啶-2'-基)喹啉锂化,然后用(i- Pr)2 PCl和(C 6 F 5)2 PCl处理,得到膦-喹啉-吡啶配体,缩写为R PQpy对于R =i- Pr和C 6 F 5。这些配体与铁和钴的二氯化物和二溴化物形成1:1加合物。FeBr 2(iPr PQpy),FeBr 2(ArFPQpy),CoCl 2(iPr PQpy),CoBr 2(iPr PQpy)和CoCl 2(ArF PQpy)证实这些配合物的M–P–C–C–N–C–C–N部分在0.078范围内为平面不同于前几代的PNN配合物,其平面度偏差约为0.35。键距和磁性表明Fe络合物为高自旋,而钴络合物为高自旋或参与自旋平衡。还研究了R PQpy配体的NNN类似物,其中膦基被异丁烯酮取代。FeBr 2(Mes
-
Synthesis of aminoalkylsilanes with oligo(ethylene oxide) unit as multifunctional electrolyte additives for lithium-ion batteries作者:JingLun Wang、Hao Luo、YongJin Mai、XinYue Zhao、LingZhi ZhangDOI:10.1007/s11426-013-4861-5日期:2013.6Aminoalkylsilanes with oligo(ethylene oxide) units were designed and synthesized as multifunctional electrolyte additives for lithium-ion batteries. The chemical structures were fully characterized by nuclear magnetic resonance (NMR) spectroscopy and their thermal properties, viscosities, electrochemical windows, and ionic conductivities were systematically measured. With adding one of these compounds (1 vol. %, DSC3N1) in the baseline electrolyte 1.0 M LiPF6 in EC: DEC (1:1, in volume), Li/LiCoO2 half cell tests showed an improved cyclability after 100 cycles and improved rate capability at 5C rate condition. Electrochemical impedance spectroscopy (EIS), X-ray photoelectron spectroscopy (XPS), and energy dispersive spectroscopic (EDS) analysis confirmed the acid scavenging function and film forming capability of DSC3N1. These results demonstrated that the multifunctional organosilicon compounds have considerable potential as additives for use in lithium-ion batteries.
-
A Strategy for the Synthesis of Well-Defined Iron Catalysts and Application to Regioselective Diene Hydrosilylation作者:Jessica Y. Wu、Benjamin N. Stanzl、Tobias RitterDOI:10.1021/ja106853y日期:2010.9.29the development of a well-defined Fe catalyst and its application to the regio- and stereoselective 1,4-hydrosilylation of 1,3-dienes. To the best of our knowledge, this is the first example of accessing a characterized low-valent Fe catalyst by controlled reductive elimination from a readily accessible Fe precatalyst.我们报告了明确定义的 Fe 催化剂的开发及其在 1,3-二烯的区域选择性和立体选择性 1,4-氢化硅烷化中的应用。据我们所知,这是第一个通过从易于获得的 Fe 预催化剂中受控还原消除来获得表征低价 Fe 催化剂的例子。
-
New Zinc Catalyst for Hydrosilylation of Carbonyl Compounds作者:Iryna D. Alshakova、Georgii I. NikonovDOI:10.1055/s-0037-1611824日期:2019.9bonds, and thus can be useful for the synthesis of allylic alcohols from α,β-unsaturated aldehydes and ketones. A new zinc complex was synthesized and applied in the catalytic hydrosilylation of carbonyl compounds. Optimization of the reaction conditions showed that the presence a substoichiometric amount of methanol accelerates the process significantly. The reaction can proceed at very low catalyst
表征谱图
-
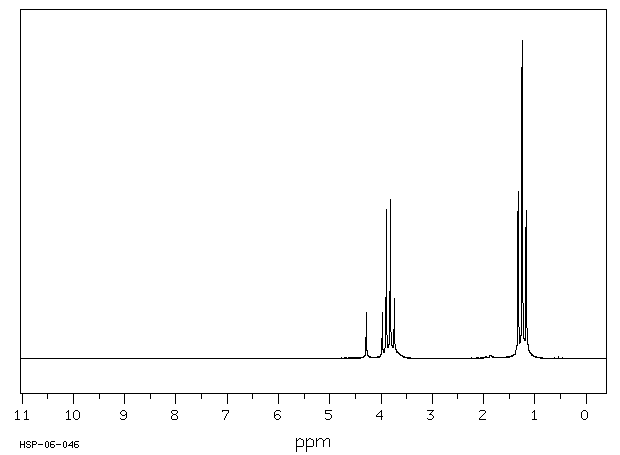
氢谱1HNMR
-
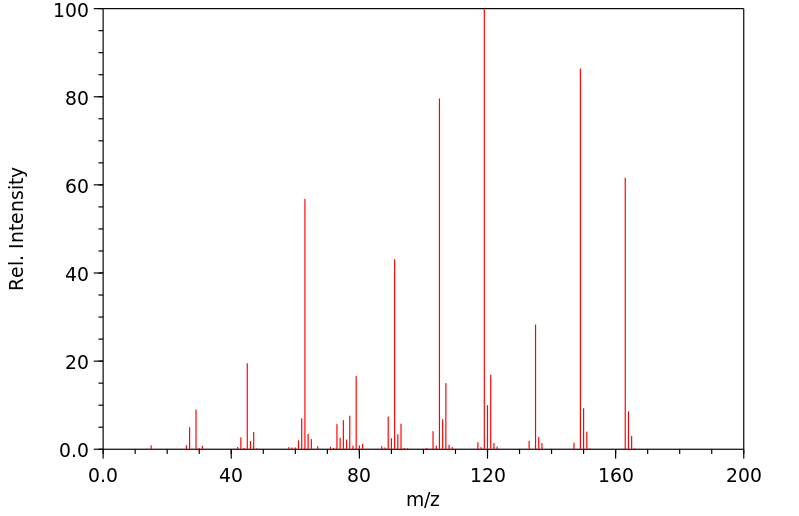
质谱MS
-
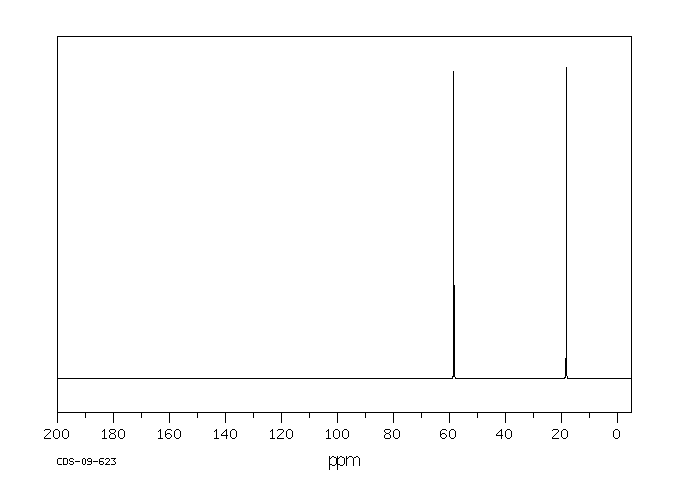
碳谱13CNMR
-
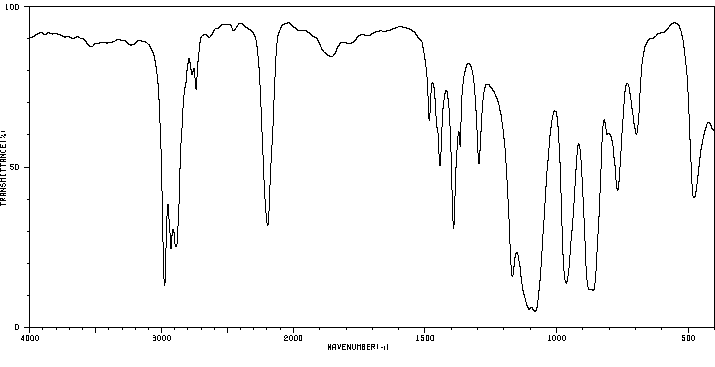
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(2-溴乙氧基)-特丁基二甲基硅烷
鲸蜡基聚二甲基硅氧烷
骨化醇杂质DCP
马沙骨化醇中间体
马来酸双(三甲硅烷)酯
顺式-二氯二(二甲基硒醚)铂(II)
顺-N-(1-(2-乙氧基乙基)-3-甲基-4-哌啶基)-N-苯基苯酰胺
降钙素杂质13
降冰片烯基乙基三甲氧基硅烷
降冰片烯基乙基-POSS
间-氨基苯基三甲氧基硅烷
镓,二(1,1-二甲基乙基)甲基-
镁,氯[[二甲基(1-甲基乙氧基)甲硅烷基]甲基]-
锑,二溴三丁基-
铷,[三(三甲基甲硅烷基)甲基]-
铂(0)-1,3-二乙烯-1,1,3,3-四甲基二硅氧烷
钾(4-{[二甲基(2-甲基-2-丙基)硅烷基]氧基}-1-丁炔-1-基)(三氟)硼酸酯(1-)
金刚烷基乙基三氯硅烷
酰氧基丙基双封头
达格列净杂质
辛醛,8-[[(1,1-二甲基乙基)二甲基甲硅烷基]氧代]-
辛甲基-1,4-二氧杂-2,3,5,6-四硅杂环己烷
辛基铵甲烷砷酸盐
辛基衍生化硅胶(C8)ZORBAX?LP100/40C8
辛基硅三醇
辛基甲基二乙氧基硅烷
辛基三甲氧基硅烷
辛基三氯硅烷
辛基(三苯基)硅烷
辛乙基三硅氧烷
路易氏剂-3
路易氏剂-2
路易士剂
试剂Cyanomethyl[3-(trimethoxysilyl)propyl]trithiocarbonate
试剂3-[Tris(trimethylsiloxy)silyl]propylvinylcarbamate
试剂3-(Trimethoxysilyl)propylvinylcarbamate
试剂2-(Trimethylsilyl)cyclopent-2-en-1-one
试剂11-Azidoundecyltriethoxysilane
西甲硅油杂质14
衣康酸二(三甲基硅基)酯
苯胺,4-[2-(三乙氧基甲硅烷基)乙基]-
苯磺酸,羟基-,盐,单钠聚合甲醛,1,3,5-三嗪-2,4,6-三胺和脲
苯甲醇,a-[(三苯代甲硅烷基)甲基]-
苯并磷杂硅杂英,5,10-二氢-10,10-二甲基-5-苯基-
苯基二甲基氯硅烷
苯基二甲基乙氧基硅
苯基二甲基(2'-甲氧基乙氧基)硅烷
苯基乙酰氧基三甲基硅烷
苯基三辛基硅烷
苯基三甲氧基硅烷