9,10-二氯蒽 | 605-48-1
中文名称
9,10-二氯蒽
中文别名
——
英文名称
9,10-dichloroanthracene
英文别名
——
CAS
605-48-1
化学式
C14H8Cl2
mdl
MFCD00001246
分子量
247.124
InChiKey
FKDIWXZNKAZCBY-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:210-216 °C
-
沸点:321.34°C (rough estimate)
-
密度:1.08
-
溶解度:可溶于氯仿(少许)、二氯甲烷(少许)
-
保留指数:2187.3
-
稳定性/保质期:
性质与稳定性:在常温常压下,该物质不会分解。
计算性质
-
辛醇/水分配系数(LogP):5.7
-
重原子数:16
-
可旋转键数:0
-
环数:3.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:0
-
氢给体数:0
-
氢受体数:0
安全信息
-
TSCA:Yes
-
危险品标志:Xi
-
安全说明:S26,S36,S37/39
-
危险类别码:R36,R36/37/38
-
海关编码:2903999090
-
储存条件:贮存: 将密器密封后,放入密封的主容器中,并存放于阴凉、干燥处。
SDS
| Name: | 9 10-Dichloroanthracene Material Safety Data Sheet |
| Synonym: | None |
| CAS: | 605-48-1 |
Synonym:None
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 605-48-1 | 9,10-Dichloroanthracene | ca 100 | 210-087-9 |
Risk Phrases: 36/37/38
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Irritating to eyes, respiratory system and skin.The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
Causes eye irritation. May cause chemical conjunctivitis.
Skin:
Causes skin irritation.
Ingestion:
May cause gastrointestinal irritation with nausea, vomiting and diarrhea. The toxicological properties of this substance have not been fully investigated.
Inhalation:
Causes respiratory tract irritation. The toxicological properties of this substance have not been fully investigated. Can produce delayed pulmonary edema.
Chronic:
Effects may be delayed.
Section 4 - FIRST AID MEASURES
Eyes: Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid. Do NOT use mouth-to-mouth resuscitation. If breathing has ceased apply artificial respiration using oxygen and a suitable mechanical device such as a bag and a mask.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion.
Extinguishing Media:
Use agent most appropriate to extinguish fire. Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container. Clean up spills immediately, observing precautions in the Protective Equipment section. Avoid generating dusty conditions.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Use with adequate ventilation. Minimize dust generation and accumulation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation.
Storage:
Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 605-48-1: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant respirator use.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Powder
Color: yellowish - gold
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 212.5 - 215.5 deg C
Autoignition Temperature: Not applicable.
Flash Point: Not applicable.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density: 1.0810g/cm3
Molecular Formula: C14H8Cl2
Molecular Weight: 247.12
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable at room temperature in closed containers under normal storage and handling conditions.
Conditions to Avoid:
Incompatible materials, dust generation, excess heat, strong oxidants.
Incompatibilities with Other Materials:
Oxidizing agents.
Hazardous Decomposition Products:
Carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, oxides of chlorine.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 605-48-1 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
9,10-Dichloroanthracene - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XI
Risk Phrases:
R 36/37/38 Irritating to eyes, respiratory system
and skin.
Safety Phrases:
S 26 In case of contact with eyes, rinse immediately
with plenty of water and seek medical advice.
S 28A After contact with skin, wash immediately with
plenty of water.
S 37 Wear suitable gloves.
S 37/39 Wear suitable gloves and eye/face
protection.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 605-48-1: No information available.
Canada
CAS# 605-48-1 is listed on Canada's NDSL List.
CAS# 605-48-1 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 605-48-1 is listed on the TSCA inventory.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
反应信息
-
作为反应物:参考文献:名称:Significant stabilization of palladium by gold in the bimetallic nanocatalyst leading to an enhanced activity in the hydrodechlorination of aryl chlorides摘要:Au对Pd的稳定作用增强了Au/Pd双金属纳米团簇中Pd的反应性,并改变了反应机制。DOI:10.1039/c5cc04432d
-
作为产物:参考文献:名称:芳烃电化学氯化和硝化的相转移催化摘要:一种独特的双相电化学反应,其中四丁基铵 (TBA + ) 盐起到电解质和相转移催化剂的双重作用,被开发用于使用廉价的无机盐进行芳香族化合物的氯化和硝化,产率高达 98%,反应时间更短,更高的法拉第效率。DOI:10.1002/anie.202319206
文献信息
-
PHOTOSENSITIVE RESIN COMPOSITION, OXIME SULFONATE COMPOUND, METHOD FOR FORMING CURED FILM, CURED FILM, ORGANIC EL DISPLAY DEVICE, AND LIQUID CRYSTAL DISPLAY DEVICE申请人:FUJIFILM Corporation公开号:US20130171415A1公开(公告)日:2013-07-04Disclosed is a photosensitive resin composition comprising: (Component A) an oxime sulfonate compound represented by Formula (1); (Component B) a resin comprising a constituent unit having an acid-decomposable group that is decomposed by an acid to form a carboxyl group or a phenolic hydroxy group; and (Component C) a solvent wherein in Formula (1) R 1 denotes an alkyl group, an aryl group, or a heteroaryl group, each R 2 independently denotes a hydrogen atom, an alkyl group, an aryl group, or a halogen atom, Ar 1 denotes an o-arylene group or an o-heteroarylene group, X denotes O or S, and n denotes 1 or 2, provided that of two or more R 2 s present in the compound, at least one denotes an alkyl group, an aryl group, or a halogen atom.
-
Aromatic substitution in ball mills: formation of aryl chlorides and bromides using potassium peroxomonosulfate and NaX作者:Robert Schmidt、Achim Stolle、Bernd OndruschkaDOI:10.1039/c2gc16508b日期:——Aryl chlorides and bromides are formed from arenes in a ball mill using KHSO5 and NaX (X = Cl, Br) as oxidant and halogen source, respectively. Investigation of the reaction parameters identified operating frequency, milling time, and the number of milling balls as the main influencing variables, as these determine the amount of energy provided to the reaction system. Assessment of liquid-assisted grinding conditions revealed, that the addition of solvents has no advantageous effect in this special case. Preferably activated arenes are halogenated, whereby bromination afforded higher product yields than chlorination. Most often reactions are regio- and chemoselective, since p-substitution was preferred and concurring side-chain oxidation of alkylated arenes by KHSO5 was not observed.
-
DIRECT ANTI-MARKOVNIKOV ADDITION OF ACIDS TO ALKENES申请人:THE UNIVERSITY OF NORTH CAROLINA AT CHAPEL HILL公开号:US20160096791A1公开(公告)日:2016-04-07A method of making an anti-Markovnikov addition product, comprises reacting an acid with an alkene or alkyne in a dual catalyst reaction system to the exclusion of oxygen to produce said anti-Markovnikov addition product; the dual catalyst reaction system comprising a single electron oxidation catalyst in combination with a hydrogen atom donor catalyst. Dual catalyst composition useful for carrying out such methods are also described.
-
Triptycenyl Sulfide: A Practical and Active Catalyst for Electrophilic Aromatic Halogenation Using <i>N</i>-Halosuccinimides作者:Yuji Nishii、Mitsuhiro Ikeda、Yoshihiro Hayashi、Susumu Kawauchi、Masahiro MiuraDOI:10.1021/jacs.9b12672日期:2020.1.22A Lewis base catalyst Trip-SMe (Trip = triptycenyl) for electrophilic aromatic halogenation using N-halosuccinimides (NXS) is introduced. In the presence of an appropriate activator (as a non-coordinating-anion source), a series of unactivated aromatic compounds were halogenated at ambient temperature using NXS. This catalytic system was applicable to transformations that are currently unachievable介绍了一种用于使用 N-卤代琥珀酰亚胺 (NXS) 进行亲电芳香卤化的路易斯碱催化剂 Trip-SMe(Trip = triptycenyl)。在合适的活化剂(作为非配位阴离子源)存在下,一系列未活化的芳香族化合物在环境温度下使用 NXS 进行卤化。该催化体系适用于目前除使用 Br2 或 Cl2 之外无法实现的转化:例如萘的多卤化、BINOL 的区域选择性溴化等。 对照实验表明,三烯基取代基对催化活性起着至关重要的作用,和动力学实验暗示锍盐 [Trip-S(Me)Br][SbF6] 作为活性物质的存在。与简单的二烷基硫醚相比,Trip-SMe 在卤络合物中表现出显着的电荷分离离子对特征,其结构信息是通过单晶 X 射线分析获得的。一项初步的计算研究表明,三烯基官能团的 π 系统是巩固亲电性增强的关键基序。
-
유기발광 화합물 및 이를 포함하는 유기전계발광소자申请人:PnH tech (주)피엔에이치테크(120100135796) Corp. No ▼ 131111-0237931BRN ▼129-86-36614公开号:KR20150124637A公开(公告)日:2015-11-06본 발명은 유기전계발광소자에 채용되는 유기발광 화합물에 관한 것으로서, 하기 [화학식 1]로 표시되는 것을 특징으로 하고, 이를 정공수송 가능층 또는 발광층 내의 인광 호스트 화합물로 채용하는 경우 구동전압, 휘도 및 장수명 등의 발광특성이 우수한 유기전계발광소자를 구현할 수 있다. [화학식 1]
表征谱图
-
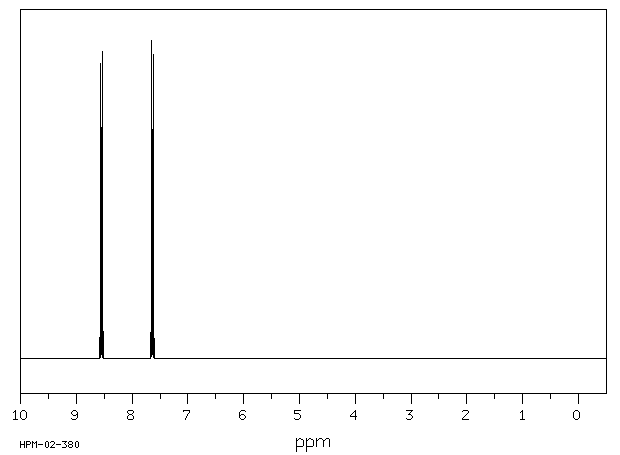
氢谱1HNMR
-
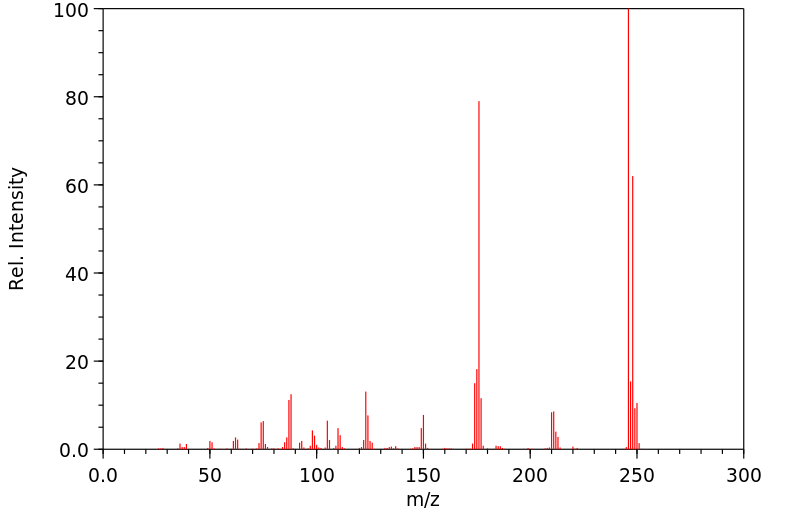
质谱MS
-
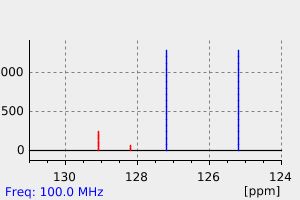
碳谱13CNMR
-
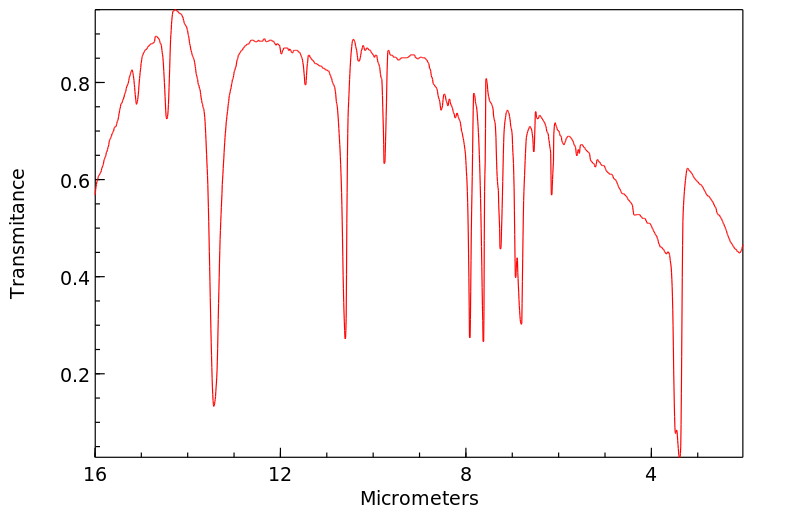
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
齐斯托醌
黄决明素
马普替林相关物质D
马普替林杂质E(N-甲基马普替林)
马普替林杂质D
马普替林D3
马普替林
颜料黄199
颜料黄147
颜料黄123
颜料黄108
颜料红89
颜料红85
颜料红251
颜料红177
颜料紫27
顺式-1-(9-蒽基)-2-硝基乙烯
阿美蒽醌
阳离子蓝FGL
阳离子蓝3RL
长蠕孢素
镁蒽四氢呋喃络合物
镁蒽
锈色洋地黄醌醇
锂钠2-[[4-[[3-[(4-氨基-9,10-二氧代-3-磺基-1-蒽基)氨基]-2,2-二甲基-丙基]氨基]-6-氯-1,3,5-三嗪-2-基]氨基]苯-1,4-二磺酸酯
锂胭脂红
链蠕孢素
铷离子载体I
铝洋红
铂(2+)二氯化1-({2-[(2-氨基乙基)氨基]乙基}氨基)蒽-9,10-二酮(1:1)
钾6,11-二氧代-6,11-二氢-1H-蒽并[1,2-d][1,2,3]三唑-4-磺酸酯
钠alpha-(丙烯酰氨基)-[4-[[9,10-二氢-4-(异丙基氨基)-9,10-二氧代-1-蒽基]氨基]苯氧基]甲苯磺酸盐
钠[[3-[[4-(环己基氨基)-9,10-二氢-9,10-二氧代-1-蒽基]氨基]-1-氧代丙基]氨基]苯磺酸盐
钠[3-[[9,10-二氢-4-(异丙基氨基)-9,10-二氧代-1-蒽基]氨基]丁基]苯磺酸盐
钠6,11-二氧代-6,11-二氢-1H-蒽并[1,2-d][1,2,3]三唑-4-磺酸酯
钠4-({4-[乙酰基(乙基)氨基]苯基}氨基)-1-氨基-9,10-二氧代-9,10-二氢-2-蒽磺酸酯
钠2-[(4-氨基-9,10-二氧代-3-磺基-9,10-二氢-1-蒽基)氨基]-4-{[2-(磺基氧基)乙基]磺酰基}苯甲酸酯
钠1-氨基-9,10-二氢-4-[[4-(1,1-二甲基乙基)-2-甲基苯基]氨基]-9,10-二氧代蒽-2-磺酸盐
钠1-氨基-4-[(3-{[(4-甲基苯基)磺酰基]氨基}苯基)氨基]-9,10-二氧代-9,10-二氢-2-蒽磺酸酯
钠1-氨基-4-[(3,4-二甲基苯基)氨基]-9,10-二氧代-9,10-二氢-2-蒽磺酸酯
钠1-氨基-4-(1,3-苯并噻唑-2-基硫基)-9,10-二氧代蒽-2-磺酸盐
醌茜隐色体
醌茜素
酸性蓝P-RLS
酸性蓝41
酸性蓝27
酸性蓝127:1
酸性紫48
酸性紫43
酸性兰62