4-异丙基苯基硫代异氰酸酯 | 89007-45-4
中文名称
4-异丙基苯基硫代异氰酸酯
中文别名
1-异丙基-4-异硫氰酸苯酯;4-异丙基苯基异硫氰酸酯
英文名称
4-isopropylphenyl isothiocyanate
英文别名
1-isopropyl-4-isothiocyanatobenzene;1-isothiocyanato-4-propan-2-ylbenzene
CAS
89007-45-4
化学式
C10H11NS
mdl
MFCD00022060
分子量
177.27
InChiKey
SNWJJTIJIDXIHL-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
计算性质
-
辛醇/水分配系数(LogP):4.7
-
重原子数:12
-
可旋转键数:2
-
环数:1.0
-
sp3杂化的碳原子比例:0.3
-
拓扑面积:44.4
-
氢给体数:0
-
氢受体数:2
安全信息
-
危险等级:6.1
-
危险品标志:Xn
-
危险类别码:R20/21/22,R36/37/38
-
危险品运输编号:2810
-
海关编码:2930909090
-
包装等级:II
-
危险类别:6.1
-
安全说明:S26,S36/37/39
-
储存条件:在干燥、阴凉且密闭的环境中,并建议置于惰性气体中保存。
SDS
| Name: | 4-Isopropylphenyl isothiocyanate 95+% Material Safety Data Sheet |
| Synonym: | |
| CAS: | 89007-45-4 |
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 89007-45-4 | 4-Isopropylphenyl isothiocyanate | 95+% | unlisted |
Risk Phrases: 20/21/22 36/37/38
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Harmful by inhalation, in contact with skin and if swallowed.
Irritating to eyes, respiratory system and skin.Moisture sensitive.
Potential Health Effects
Eye:
Causes eye irritation. Lachrymator (substance which increases the flow of tears).
Skin:
Causes skin irritation. Harmful if absorbed through the skin.
Ingestion:
Harmful if swallowed. May cause irritation of the digestive tract.
Inhalation:
Harmful if inhaled. Causes respiratory tract irritation.
Chronic:
Not available.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:
Get medical aid. Wash mouth out with water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media:
Use foam, dry chemical, or carbon dioxide.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Absorb spill with inert material (e.g. vermiculite, sand or earth), then place in suitable container.
Section 7 - HANDLING and STORAGE
Handling:
Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes. Use only in a chemical fume hood.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Store under an inert atmosphere.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 89007-45-4: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Liquid
Color: yellow
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 154 - 155 deg C @20mmHg
Freezing/Melting Point: Not available.
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density: 1.03
Molecular Formula: C10H11NS
Molecular Weight: 177.27
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Not available.
Conditions to Avoid:
Incompatible materials, exposure to moist air or water.
Incompatibilities with Other Materials:
Strong oxidizing agents, acids, bases.
Hazardous Decomposition Products:
Carbon monoxide, oxides of nitrogen, oxides of sulfur, carbon dioxide, acrid smoke and fumes.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 89007-45-4 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
4-Isopropylphenyl isothiocyanate - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Shipping Name: TOXIC LIQUID, ORGANIC, N.O.S.*
Hazard Class: 6.1
UN Number: 2810
Packing Group: III
IMO
Shipping Name: TOXIC LIQUID, ORGANIC, N.O.S.
Hazard Class: 6.1
UN Number: 2810
Packing Group: III
RID/ADR
Shipping Name: TOXIC LIQUID, ORGANIC, N.O.S.
Hazard Class: 6.1
UN Number: 2810
Packing group: III
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XN
Risk Phrases:
R 20/21/22 Harmful by inhalation, in contact with
skin and if swallowed.
R 36/37/38 Irritating to eyes, respiratory system
and skin.
Safety Phrases:
S 26 In case of contact with eyes, rinse immediately
with plenty of water and seek medical advice.
S 36/37/39 Wear suitable protective clothing, gloves
and eye/face protection.
WGK (Water Danger/Protection)
CAS# 89007-45-4: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 89007-45-4 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 89007-45-4 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 4-异丙基苯胺 4-Isopropylaniline 99-88-7 C9H13N 135.209
反应信息
-
作为反应物:描述:参考文献:名称:2-硝基肉桂醛衍生的缩氨基硫脲脲酶抑制剂的合成及生物学评价摘要:脲酶的过度活跃诱发消化性溃疡和胃炎的发病机制。可以通过引入各种抑制剂来降低脲酶的过量。高脲酶含量的风险也存在于农业领域,它会降低环境健康。这种令人担忧的情况导致了对这种必须在药物化学领域创造奇迹的新型支架的强制性探索。因此,我们在此报告了一系列新的N 4 -用缩氨基硫脲3a-q取代肉桂醛作为脲酶抑制的潜在候选物。这些新的N 4取代的缩氨基硫脲3a-q的模糊化学支架通过先进的光谱技术(如 ESI-MS)进行了表征,13 CNMR、1 HNMR 和 FTIR。所有新合成的化合物都显示出显着的脲酶抑制作用,IC 50值在 11.4 ± 0.23 至 80.6 ± 0.52 μM 的范围内。此外,化合物3o的动力学研究显示竞争性抑制,ki 值为 8.65 ± 0.37 µM。为了分析所有化合物的结构-活性关系和相互作用模式,使用分子对接,揭示了化合物与活性位点残基的良好结合相互作用,例如镍原子配位、π-阳离子、π-π相互作用和氢键DOI:10.1016/j.molstruc.2023.135387
-
作为产物:描述:C10H13NS2*C6H15N 在 sodium persulfate 、 potassium carbonate 作用下, 以 水 为溶剂, 反应 1.0h, 以90%的产率得到4-异丙基苯基硫代异氰酸酯参考文献:名称:Na 2 S 2 O 8介导的水中伯胺高效合成异硫氰酸酯摘要:我们已经开发了两种绿色,实用且有效的方法,包括一锅法,可通过胺和二硫化碳通过胺合成异硫氰酸酯。用过硫酸钠脱硫。使用水作为溶剂。为了使异硫氰酸酯具有良好的化学选择性,必须具备碱性条件。通过两种方法,结构令人满意的直链和支链烷基胺和芳基胺很容易以令人满意的产率转化为异硫氰酸酯。卤素,苄基CH键,甲硫基,硝基,酯,烯基,富电子或不足的(杂)芳基,乙炔基,甚至酚和醇羟基均被很好地耐受。在水中一锅法也可用于从手性胺制备手性异硫氰酸酯,以及用游离氨基修饰生物活性结构。在大规模制备中,开发了独立于柱色谱法的简单实用的纯化方法。DOI:10.1039/c8gc02261e
文献信息
-
Design and development of some phenyl benzoxazole derivatives as a potent acetylcholinesterase inhibitor with antioxidant property to enhance learning and memory作者:Pavan Srivastava、Prabhash Nath Tripathi、Piyoosh Sharma、Sachchida Nand Rai、Surya Pratap Singh、Rakesh K. Srivastava、Sharmila Shankar、Sushant K. ShrivastavaDOI:10.1016/j.ejmech.2018.11.049日期:2019.2Gaussian-based quantitative structure-activity relationship (QSAR) and virtual screening (VS) processes, some promising acetylcholinesterase inhibitors (AChEIs) having antioxidant potential were designed synthesized, characterized, and evaluated for their ability to enhance learning and memory. The synthesized phenyl benzoxazole derivatives exhibited significant antioxidant potential and AChE inhibitory activity基于基于高斯的定量结构-活性关系(QSAR)和虚拟筛选(VS)过程,设计,合成了一些有前途的具有抗氧化潜力的乙酰胆碱酯酶抑制剂(AChEI),对其进行了表征,并评估了它们增强学习和记忆的能力。合成的苯基苯并恶唑衍生物表现出显着的抗氧化潜力和AChE抑制活性,而观察到的化合物34(49.6%)的抗氧化潜力显着优于标准多奈哌齐(<10%)并与抗坏血酸(56.6%)平行。最有效的化合物34(AChE IC 50的酶动力学研究 = 0.363±0.017μM; Ki = 0.19±0.03μM)揭示了AChE抑制作用的真实性质和竞争性类型。进一步评估了化合物34的体内和离体研究结果,结果表明,与标准药物多奈哌齐相比,剂量为5 mg / kg时,认知缺陷和抗氧化潜能的显着逆转。
-
Synthesis and Biological Evaluation of Arylthiourea Derivatives with Antitubercular Activity作者:Rusong Luo、Tuomo Laitinen、Liyan Teng、Tapio Nevalainen、Maija Lahtela-Kakkonen、Baofu Zheng、Honghai Wang、Antti Poso、Xuelian ZhangDOI:10.2174/1570180811310070012日期:2013.6.1Tuberculosis (TB) is a contagious disease caused by Mycobacterium tuberculosis (M. tuberculosis), and remains one of the most life-threatening plagues for public health in the world. The emergence of drug resistant strains of TB and co-infection with HIV has further complicated TB treatment. Here, the synthesis and characterizaton of a series of compounds were described, and these were followed by evaluating for their antibacterial activity against M. tuberculosis. Several novel arylthiourea derivatives exhibited excellent activity (lowest MIC=0.09 μg/ml) against M. tuberculosis including drug resistant strains of M. tuberculosis. The results suggest that these compounds are promising candidates for new anti-TB agent development.
-
Benzoxazolamines and Benzothiazolamines: Potent, Enantioselective Inhibitors of Leukotriene Biosynthesis with a Novel Mechanism of Action作者:Edward S. Lazer、Clara K. Miao、Hin-Chor Wong、Ronald Sorcek、Denice M. Spero、Alex Gilman、Kollol Pal、Mark Behnke、Anne G. GrahamDOI:10.1021/jm00033a008日期:1994.4phenylalanine with a cyclohexyl group greatly enhance potency. Several ester bioisosteres that retain potency and enantiomeric selectivity are described. Lead optimization culminated in (S)-N-[2-cyclohexyl-1-(2-pyridinyl)ethyl]-5-methyl-2-benzoxazolamine+ ++ (43b), IC50 0.001 microM. The compounds described are not inhibitors of 5-lipoxygenase but, rather, act at the level of arachidonic acid release.
-
Compounds Which Modulate The CB2 Receptor申请人:Berry Angela公开号:US20100009964A1公开(公告)日:2010-01-14Compounds of formula (I) are disclosed. Compounds according to the and are useful for treating inflammation. Those compounds which are agonists are additionally useful for treating pain.公式(I)的化合物已被披露。根据该公式的化合物对治疗炎症有用。那些是激动剂的化合物还额外适用于治疗疼痛。
-
Synthesis and Pesticidal Properties of Thio and Seleno Analogs of Some Common Urea Herbicides作者:Jerzy Zakrzewski、Maria KrawczykDOI:10.1080/10426500802391692日期:2009.7.13Thio and seleno analogs of fenuron, isoproturon, chlorotoluron, metoxuron, monuron, and diuron were synthesized from the corresponding aryl amines. Their reaction with thiophosgene leads to isothiocyanates. Aryl amines were also converted (via isocyanides) to isoselenocyanates. The reaction of both isothio- and isoselenocyanates with dimethylamine affords the corresponding thio and seleno analogs of
表征谱图
-
氢谱1HNMR
-
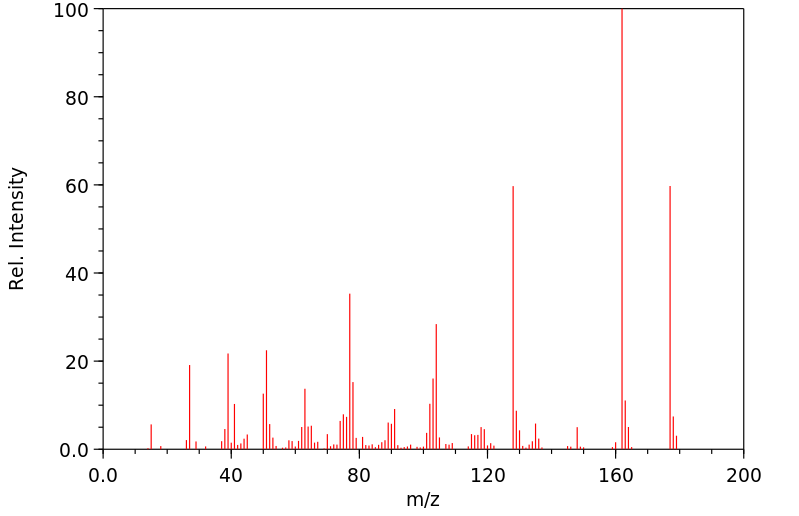
质谱MS
-
碳谱13CNMR
-
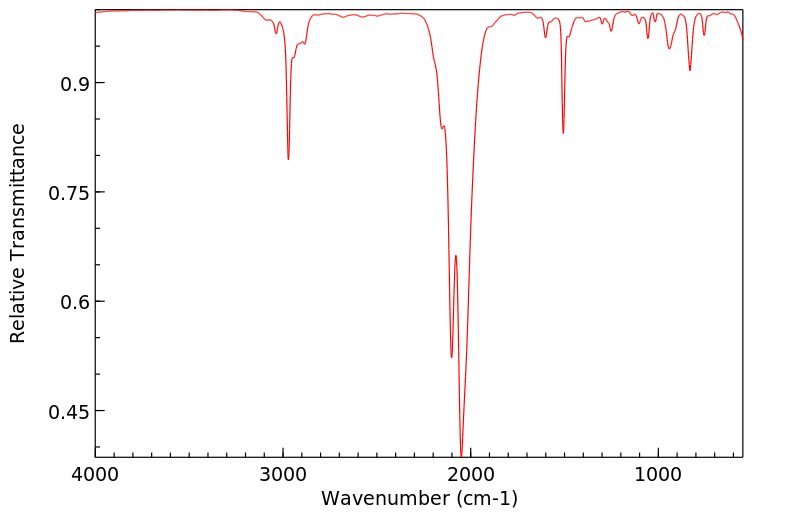
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫